Perforation Type 3 Wire – Case 3
Clinical Presentation
- 87-year-old female who presented with chest pain (CCS Class II) and was referred for staged PCI of the LAD.
Past Medical History
- HTN, HLD, DM, CAD s/p PCI, Moderate AS, Moderate to Severe MR, Pulmonary HTN
- LVEF 55%
Clinical Variables
- Prior Cardiac Catheterization: S/p PCI of the proximal and distal LCX.
Medications
- Home Medications: Aspirin, Clopidogrel, Rosuvastatin, Metoprolol Succinate, Isosorbide Mononitrate, Lisinopril
- Adjunct Pharmacotherapy: Clopidogrel, Bivalirudin
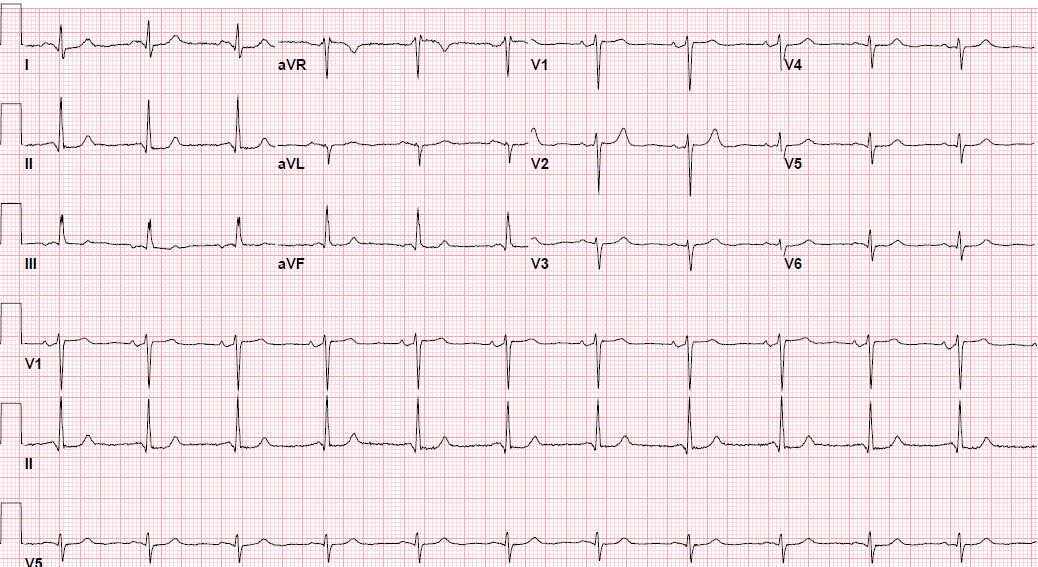
Pre-procedure EKG
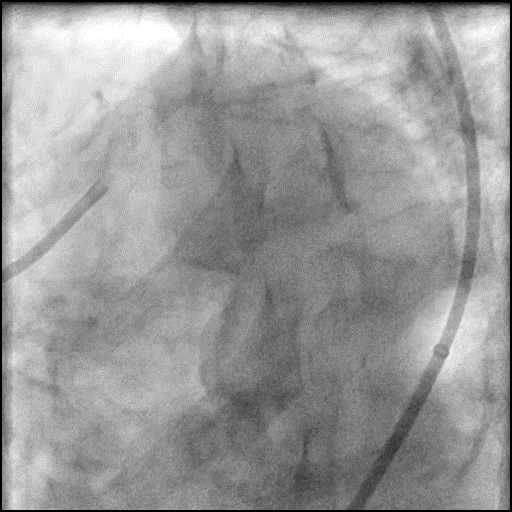
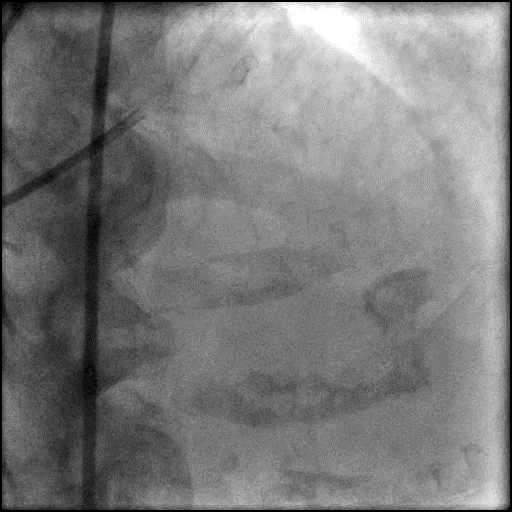
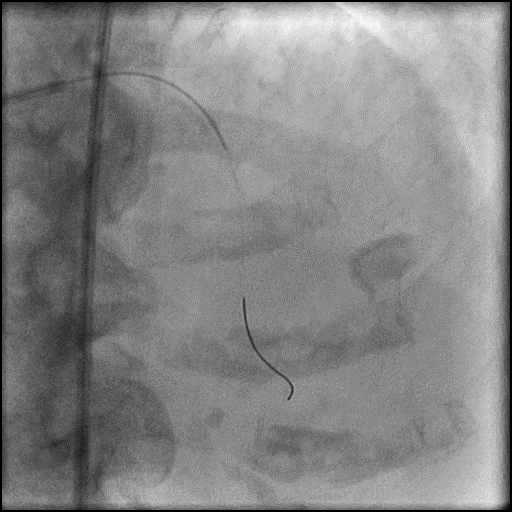
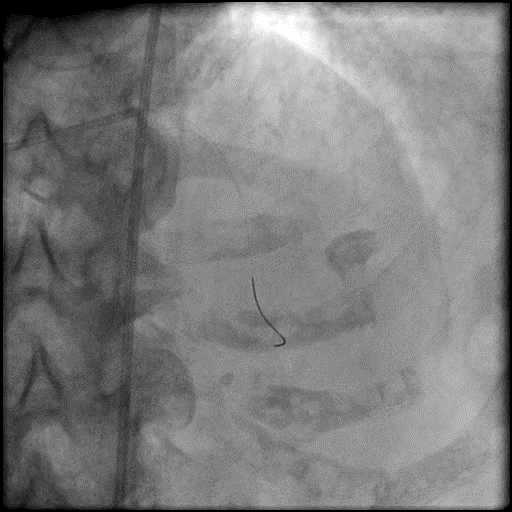
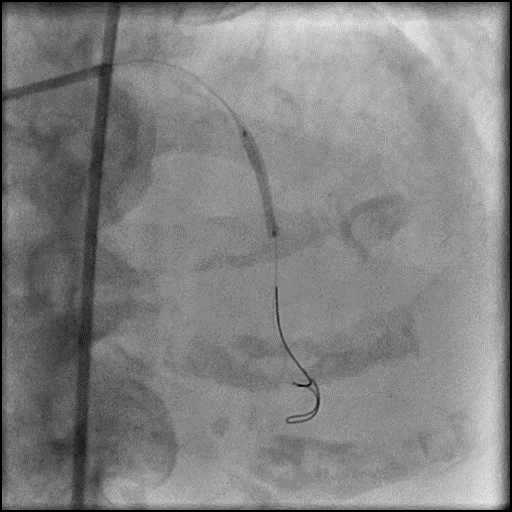
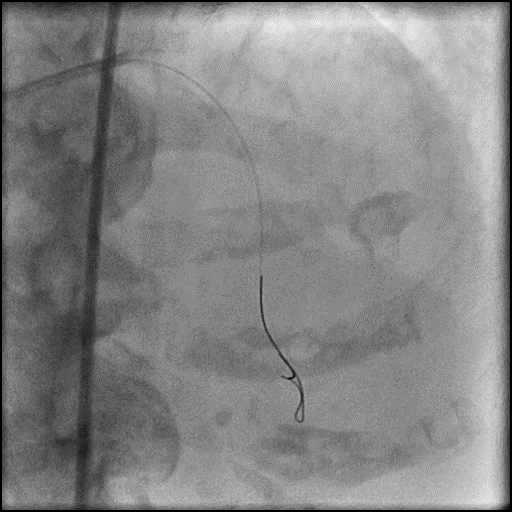
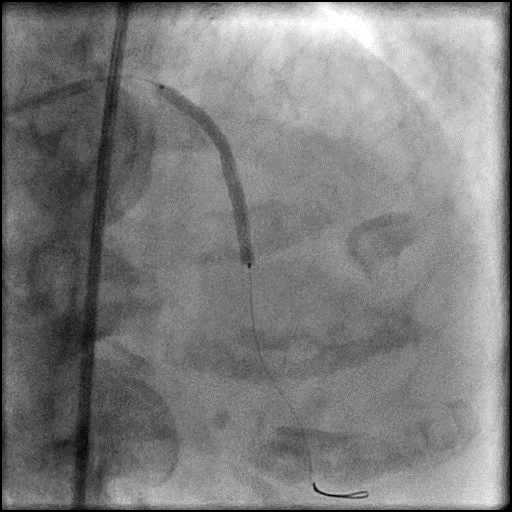
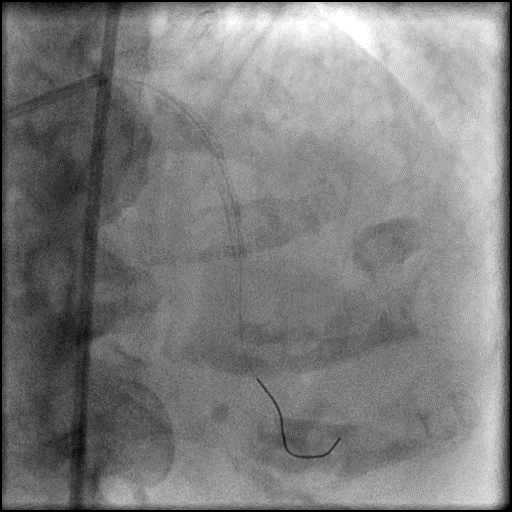
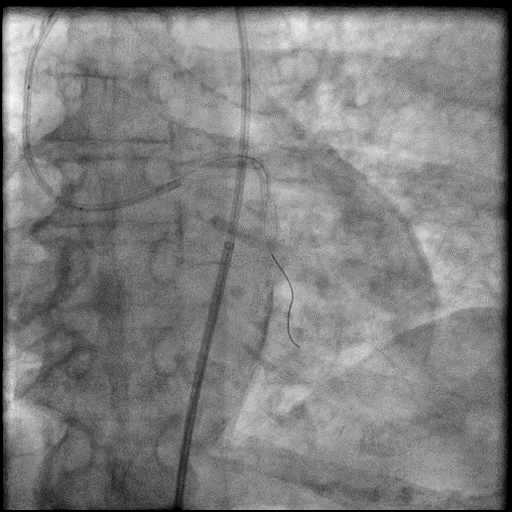
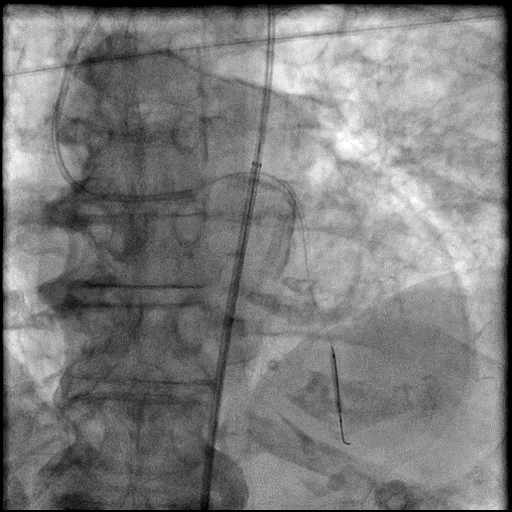
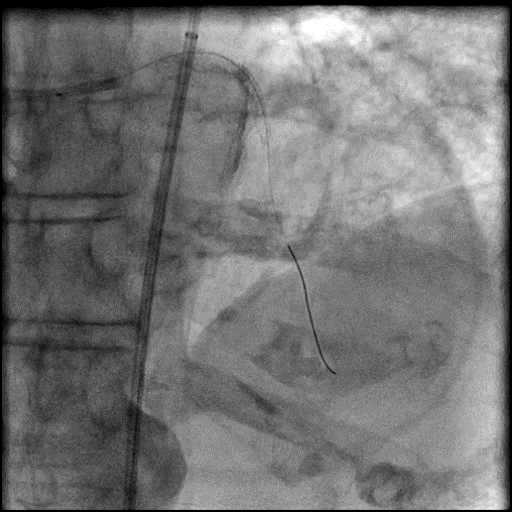
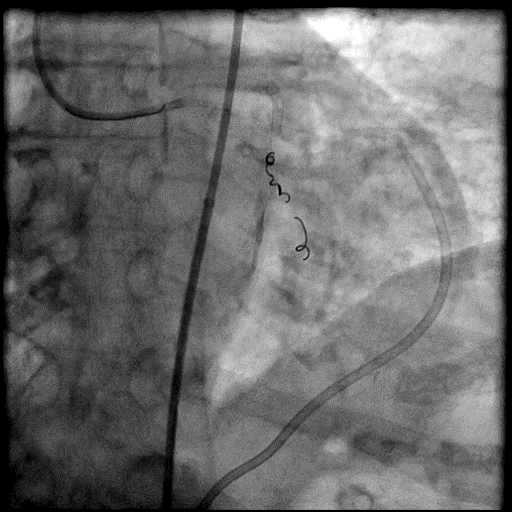
Angiograms
Post-procedure EKG
Case Overview
- Underwent intervention of the LAD.
- Procedure was complicated by a type 3 distal wire perforation of the LAD.
- Perforation was inadequately sealed with prolonged balloon tamponade of the vessel.
- Three coils were placed successfully sealing the perforation.
- Patient became hemodynamically unstable towards the end of the procedure.
- Echocardiography showed presence of a moderate size pericardial effusion with tamponade physiology.
- An emergent echocardiography guided pericardiocentesis was performed with removal of 500 cc of fluid.
- Troponin-I peaked at 6.1 ng/mL and CK-MB peaked at 16.8 ng/mL.
- Patient had a prolonged hospital course due to unrelated cardiac events, and was discharged after 14 days.
Learning Objectives
- What is the likely explanation or reason why the complication occurred?
- Wire related perforation (Around 50% of coronary perforations are guide wire related).
- How could the complication have been prevented?
- Careful manipulation of the wire. Ideally, a wire should be positioned distal to a lesion in a large caliber vessel so long as it provides sufficient support to perform a coronary intervention.
- Extreme caution needs to be taken when using a hydrophilic wire (i.e. Fielder, Whisper wires etc.). Hydrophilic wires are more prone to cause a perforation as they tend to navigate into smaller branch vessels and provide less tactile feedback.
- Using a workhorse, non-hydrophilic wire with tip load <1g when performing a coronary intervention helps reduce complications.
- Is there an alternate strategy that could have been used to manage the complication?
- Ellis Type 1 and 2 perforations usually seal spontaneously and are conservatively managed. Such patients should be closely monitored in the catheterization lab, and serial echocardiography should be performed, particularly if there is an Ellis Type 2 coronary perforation because it may lead to cardiac tamponade. Ellis Type 3 perforations are associated with increased risk of cardiac tamponade and mortality, and require immediate intervention/treatment. Ellis Type 3 Cavity Spilling perforation management is unclear. Usually they are conservatively managed, unless there is significant extravasation or the patient is symptomatic.
- Coronary perforation management algorithm:
- 1st: Prolonged balloon inflation: Position the balloon (or stent-balloon post stent deployment) just proximal or at the level of the perforation to prevent ongoing extravasation and development of hemo-pericardium. Ideally, the balloon to artery ratio should be 1:1. Inflate for 5-10 minutes followed by test deflations with contrast given in between inflations to evaluate the status of the perforation. If there is ongoing extravasation, re-inflate the balloon to stop further extravasation of blood into the pericardial space. This strategy helps stabilize the patients and gain control of the situation, while the operator prepares for echocardiography, pericardiocentesis, and more definitive treatment to seal the perforation.
- 2nd: Anticoagulation management: ‘STOP’ all anticoagulation immediately if you suspect or visualize a perforation. We consider ‘REVERSING’ heparin with protamine sulfate (to achieve ACT <225s) after coronary equipment is removed to prevent thrombosis within the vessel. If using bivalirudin, it can take up to 1-2 hours for its anticoagulation effect to a normalize after it is stopped. If patient was on glycoprotein IIB/IIIA inhibitors: For abciximab, consider giving platelet transfusion; tirofiban and eptifibatide have a short half life and their reversal can typically be achieved by stopping there infusion or in extreme cases with hemodialysis. Cangrelor has a short half life and its reversal can be achieved by stopping its infusion.
- 3rd: Covered stent: Standard of care for a perforation located in the proximal to mid segment of a vessel of appropriate size (≥2.5 mm), with no major side branch across the region where the stent will be placed. If a covered stent can be delivered to a distal vessel perforation, and the vessel is of appropriate size, covered stent placement to seal the perforation is reasonable. If the clinical situation allows, proceed with direct stent placement whenever possible using a single catheter or two-catheter (Ping-Pong) strategy. The stent should be quickly positioned and immediately deployed to high pressure. This should be followed by high pressure post-dilatation (18-20 atm) to achieve appropriate stent apposition.
- 4th: Embolization of distal vessel perforations: Non-surgical techniques for distal vessel embolization include: Coils, Gel Foams, Glues, Microspheres, Thrombin injection, Subcutaneous tissue, Autologous Blood Clots and multiple other agents (depending on what is available in an individual catheterization lab). Embolization leads to loss of vessel flow beyond point where embolized material is delivered and subsequent infarct in the vessel territory.
- 5th: Surgery Intervention: Ligation or suturing of the vessel for hemostasis with bypass grafting to the distal vessel. Pericardial patch/Teflon with possible bypass grafting to the distal vessel (consider this approach if vessel has multiple stents and/or presence of a subepicardial hematoma).
- What are the important learning points?
- Coil placement should be limited to perforations in small distal vessels as they cause complete occlusion of the vessel by thrombosis, resulting in a myocardial infarction.
- When selecting a coil, it should be slightly larger than the target vessel so it adheres with the vessel and results in successful distal vessel embolization.
- An operator needs to be very cautious when delivering a coil and assure that it does not occlude a proximal large vessel (coil selected is to large) or migrate to distal (coil selected is to small) by mistake.
- There are multiple coils available in the market and an operator should be familiar with the equipment available in their catheterization lab.
- Most commercially available coils can be delivered with a 0.018’’ microcatheter (i.e. Renegade). However, there are smaller coils available which can be delivered with a 0.014’’ microcatheter (i.e FineCross) or over the wire balloon.
- When using detachable coils, optimal positioning can be confirmed before releasing.
- When using pushable coils, they are smaller but repositioning is not possible.
- The placement of the coils should start across the perforation into the native coronary artery. Coil delivery to the site of perforation require use of a microcatheter or over the wire balloon. If using a microcatheter to deliver the coil, after the coil has been delivered, a gentle contrast injection should be performed to assess if there is any residual extravasation and if there is, then another coil should be placed. After successful sealing is confirmed, anticoagulation could be stopped/reversed after removing the intracoronary equipment (not before because of the risk of developing proximal thrombosis while performing the intervention).