Coronary Stents
The angioplasty procedures were initially performed without stent deployment, a technique that is now referred to as plain old balloon angioplasty (POBA). However, elastic recoil occurred in 5–10% of patients immediately (minutes-hours) after the procedure leading to a rebound occlusion of the artery, which often led to severe complications, including acute myocardial infarction (AMI) and the need for emergency coronary artery bypass grafting (CABG).
Coronary angioplasty with stenting has revolutionized coronary artery disease treatment, providing a scaffold to the balloon-dilated artery and prevents propagation of the dissection flaps and late recoil. The majority of PCI procedures performed currently involve balloon angioplasty and stent deployment.
The coronary stents can be categorized into two groups – Bare Metal Stent (BMS) and Drug-Eluting Stent (DES). As BMS is associated with ~ 20–30% incidence of ISR, DES is introduced to alleviate the shortcomings of BMS. [1] The majority of DES consists of a metallic alloy stent, a polymer coating (which may be durable or bioabsorbable), and an anti-restenotic drug that is mixed within the polymer and released over a period of weeks to months after implantation to lessen the local proliferative healing response.
Classification of Coronary Stents
- Bare metal stents
- Drug-eluting stents
- First generation
- Second and newer generations
- Covered Stents
- Bifurcated Stent
Drug Eluting Stents
Among multiple different types of stent available, it is challenging to differentiate which stent is superior to others.
However, some of the stents have their perspective advantages in certain scenarios.
- Resolute ONYX DES: indicated for high bleeding risk patients and labeled for 1-month DAPT. Based on the results from the Onyx ONE Clear Analysis. [2]
- Synergy XD: Due to its thinner struct with abluminal coating, it offers better trackability and pushability through various challenging anatomies. Also, available in 48 mm length.
- Ultimaster Tansei: It is a Sirolimus Eluting Cobalt Chromium system with bioresorbable polymer coating Poly DL-lactide-co-caprolactone (PDLLA /PCL) and targeted abluminal drug delivery. Expansion up to 5.8 mm (from 4.0 DES).
Covered Stent
- Graftmaster Rx (Abbott)
- PK Papyrus
- Begraft
Graftmaster Rx (Abbott)
- Effectiveness of this device for this use has not been demonstrated. Long-term outcome for this permanent implant is unknown at present.
- Minimal requirement for guide catheter size is 6, 7F
PK Papyrus (Biotronik)
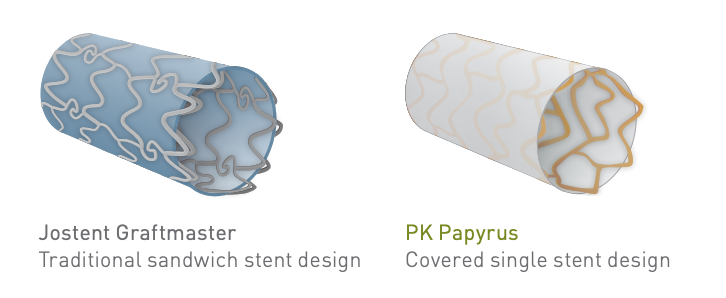
- Made of ultrathin strut stent platform (Cobalt Chromium) and has thinner profile with exceptional deliverability, compared to the graftmaster
- Indicated for the treatment of acute perforations of native coronary arteries and coronary bypass grafts in vessels 2.5 to 5.0 mm in diameter
- Compatible with 5F and 6F guide catheter
Begraft (Bentley InoMed GmbH)
- It is a cobalt-chrome stent platform, covered with a micro-porous ePTFE membrane
- Compatible with 5F guide catheter for all sizes
Bifurcated Stent
- Hoffmann R, Mintz GS, Dussaillant GR, et al. Patterns and mechanisms of in-stent restenosis. A serial intravascular ultrasound study, Circulation, 1996, vol. 94 (pg. 1247-54)10.1161/01.CIR.94.6.1247
- Windecker S, Latib A, Kedhi E, et al., on behalf of the ONYX ONE Investigators. Polymer-based or Polymer-free Stents in Patients at High Bleeding Risk. N Engl J Med 2020;382:1208-18.