Case 21: NIRS-IVUS guided PCI
Case Presentation
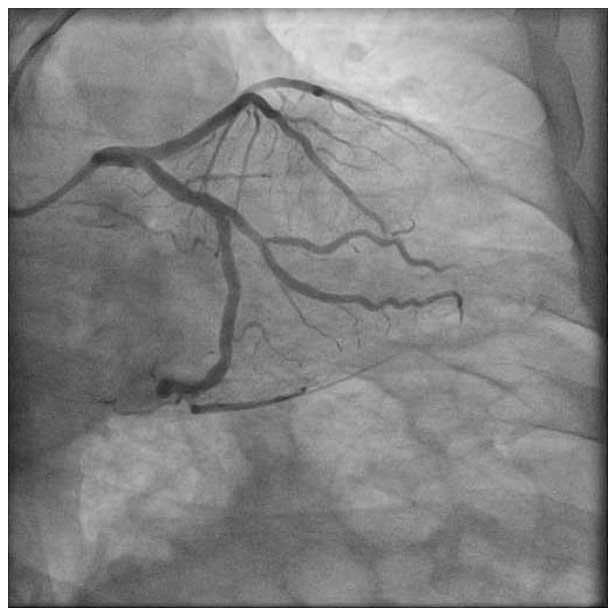
A 52-year-old female presented with CCS class III chest pain to our hospital. The patient had a history of hypertension, diabetes mellitus, and hyperlipidemia. A coronary angiography showed a 80-90% stenosis in the OM1.
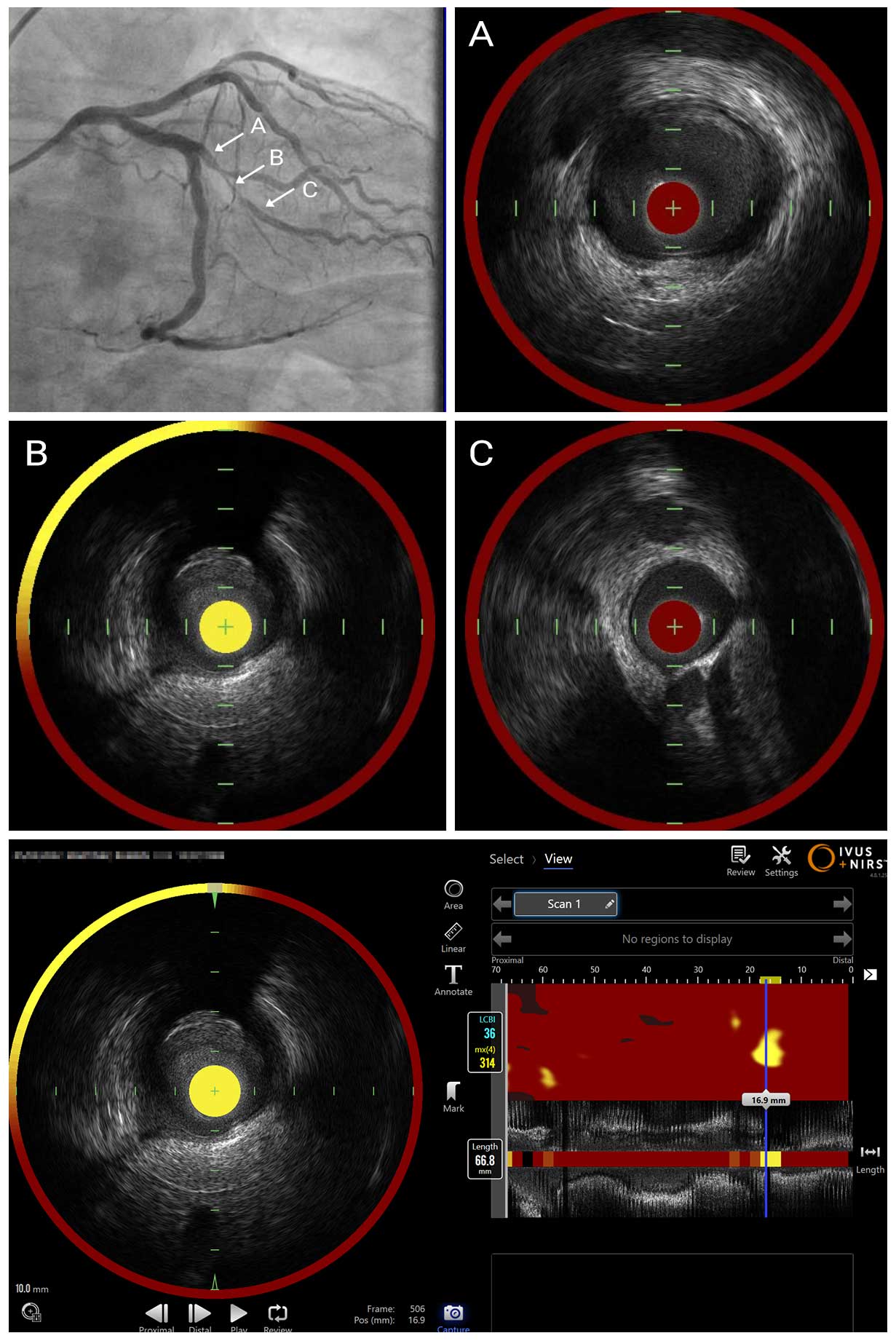
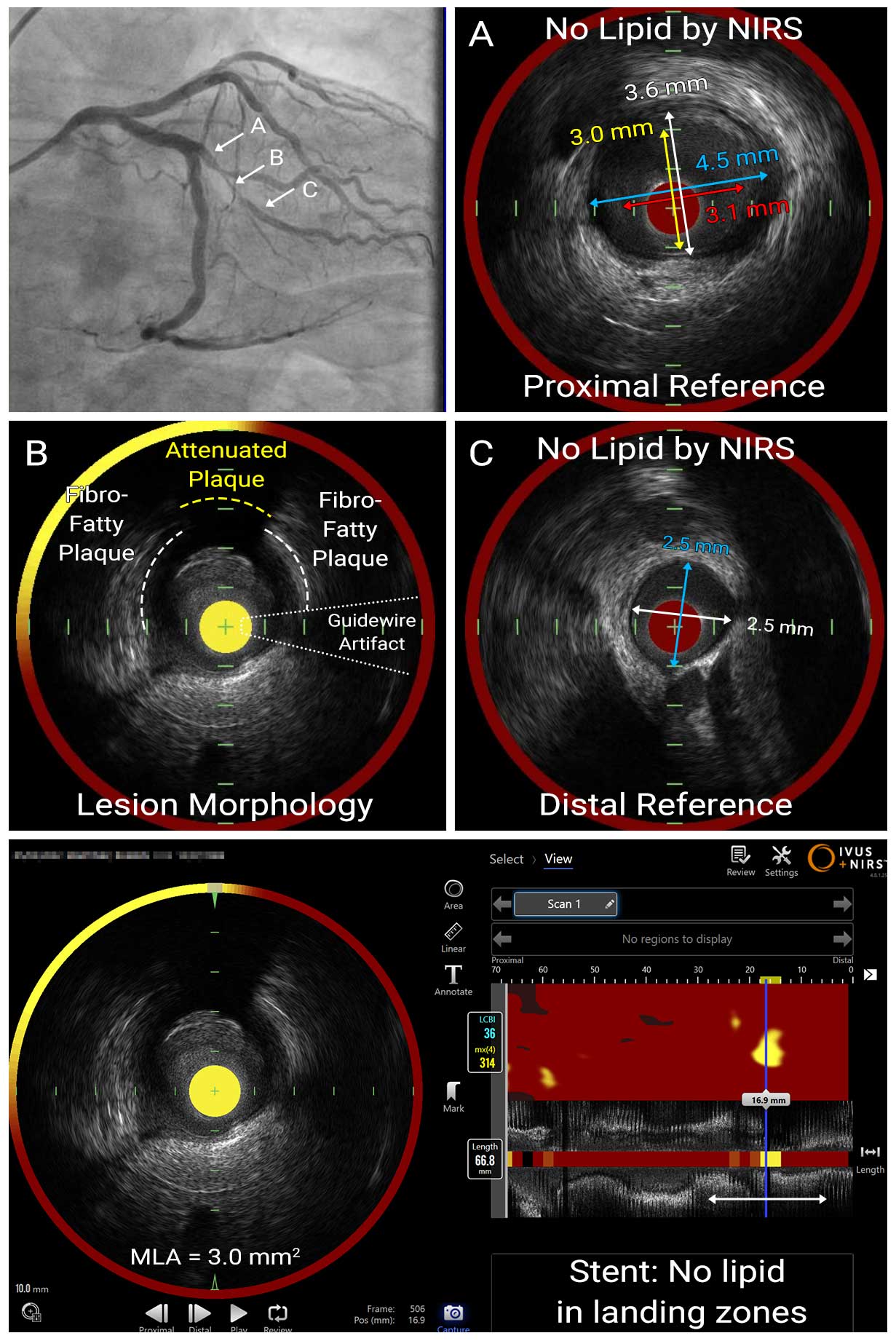
IVUS Pullback with NIRS Overlay
Pre-interventional intravascular imaging assessment was performed with a combined NIRS-IVUS catheter. IVUS identified fibro-fatty lesion with the MLA was 3.0mm². NIRS chemogram provided quantitative metrics of the lipid: the maximum lipid core burden index in a 4mm segment (maxLCBI4mm) was 314 and lesion LCBI was 36, confirming high lipid content in the lesion. IVUS-NIRS was used for stent sizing to select reference segments with small plaque burden on IVUS and no lipid by NIRS. The average diameter at the distal reference was 2.5mm (EEL-EEL) and lesion length was 17.5mm by IVUS. Based on these findings, we performed pre-dilatation with 2.5mm NC balloon, deployed 2.5 x 24mm DES, and performed post-dilatation with 3.0mm NC balloon.
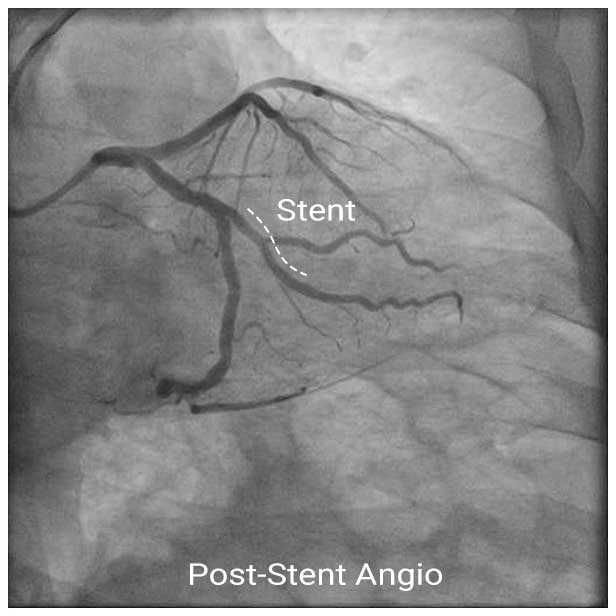
Pre/Post Angios
In this case, IVUS enabled appropriate stent sizing and stent placement. NIRS was also used to quantitatively measure lipid plaque and to use lipid-free areas as landing zones (proximal reference, distal reference) for the stent.