Dissection Type F – Case 1
Clinical Presentation
- 86-year-old female who presented with chest pain to an outside hospital and underwent cardiac catheterization showing severe 3-Vessel CAD with LM involvement. She was referred for complex PCI.
Past Medical History
- HTN, HLD, CAD, AAA s/p Repair, Carotid Stenosis (Right Total Occlusion, 60% Stenosis on Left), Moderate AS, COPD on 2L O2 NC at Home
Clinical Variables
- Prior Cardiac Catheterization: LM 70-80% stenosis, and RCA 80-90% stenosis.
Medications
- Home Medications: Clopidogrel, Metoprolol Tartrate, Amlodipine, Furosemide, Albuterol, Tiotropium
- Adjunct Pharmacotherapy: Clopidogrel, Bivalirudin
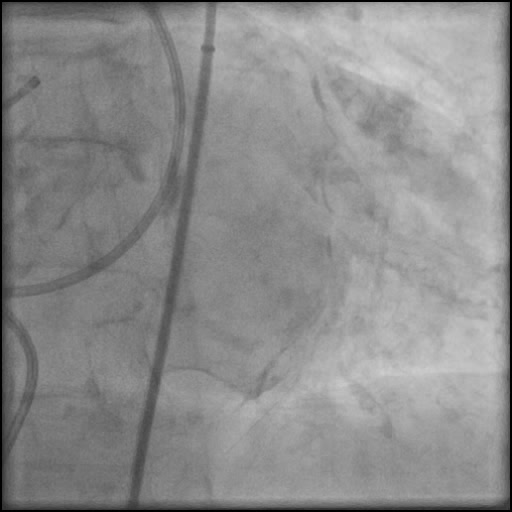
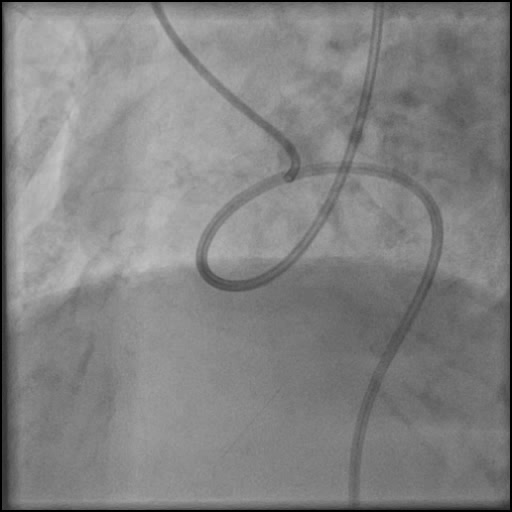
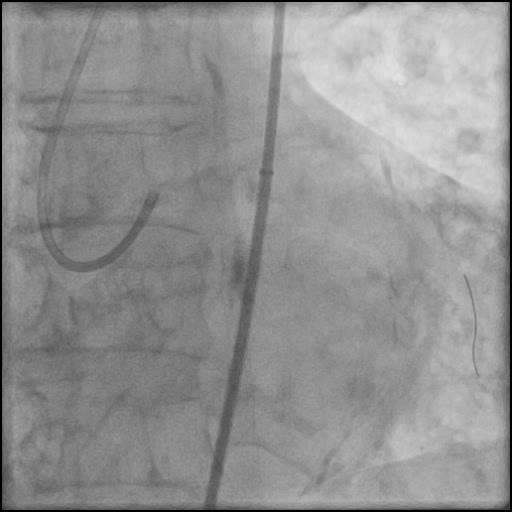
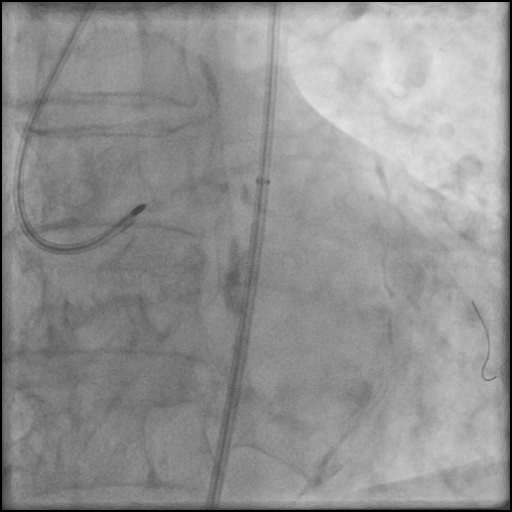
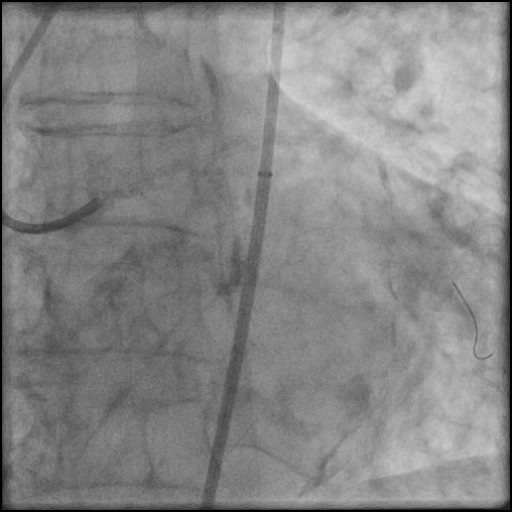
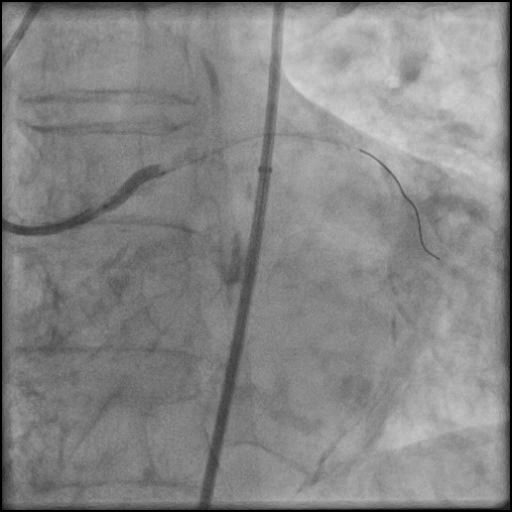
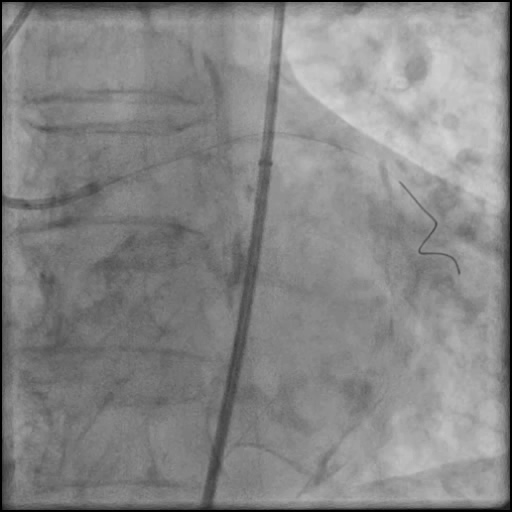
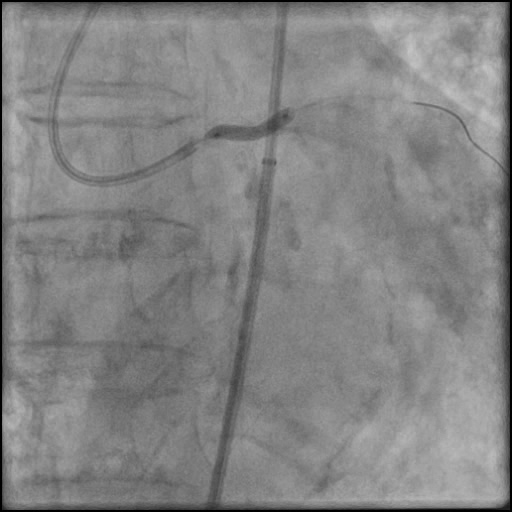
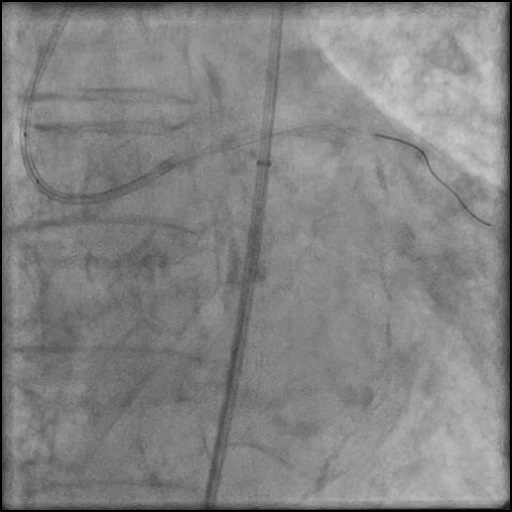
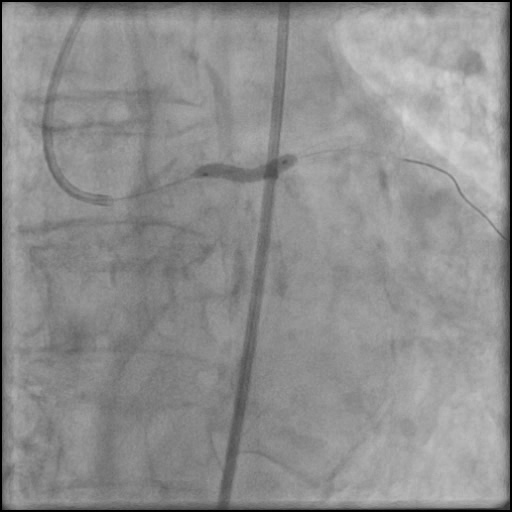
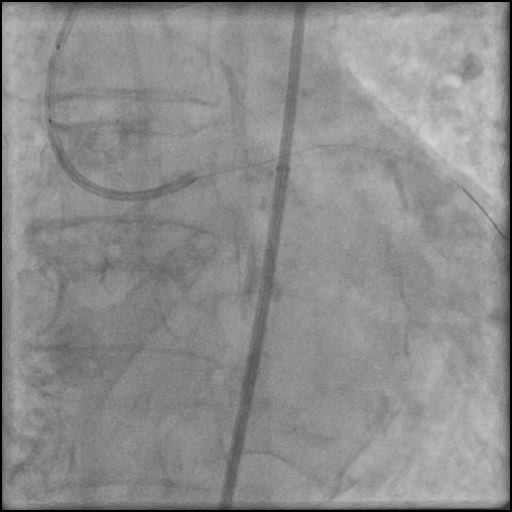
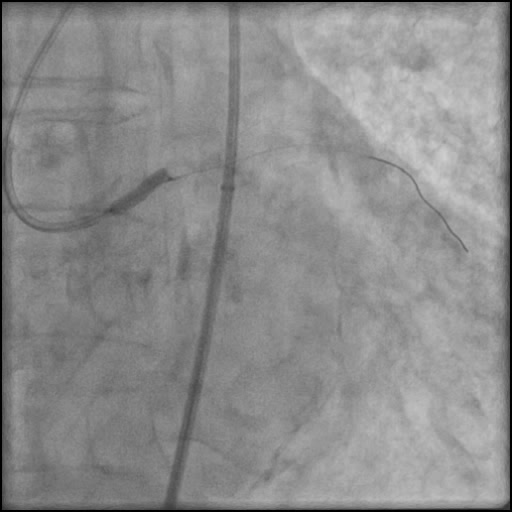
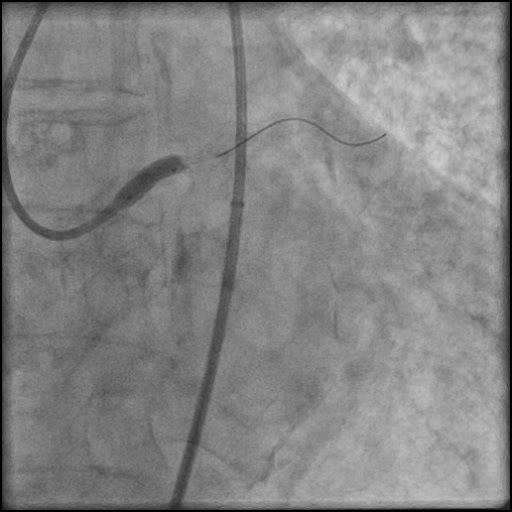
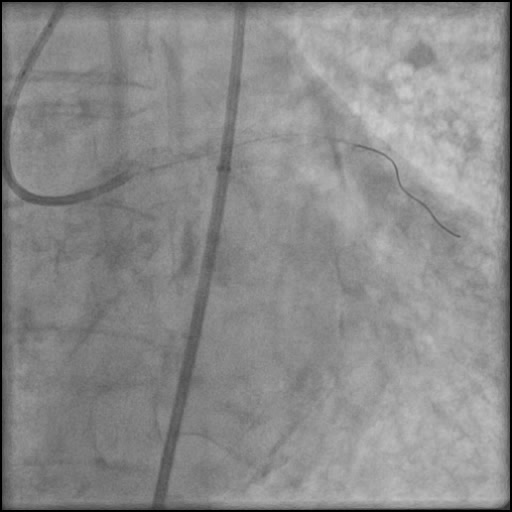
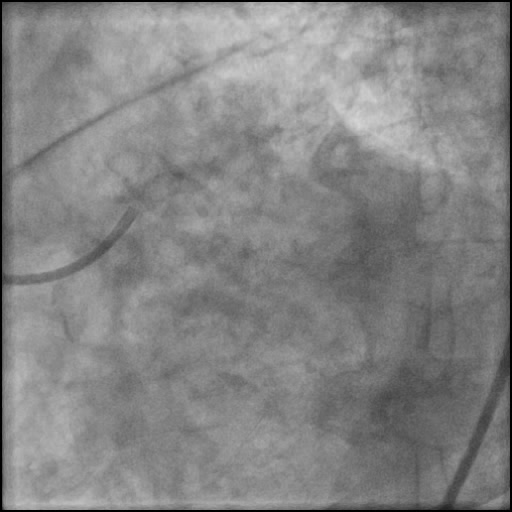
Angiograms
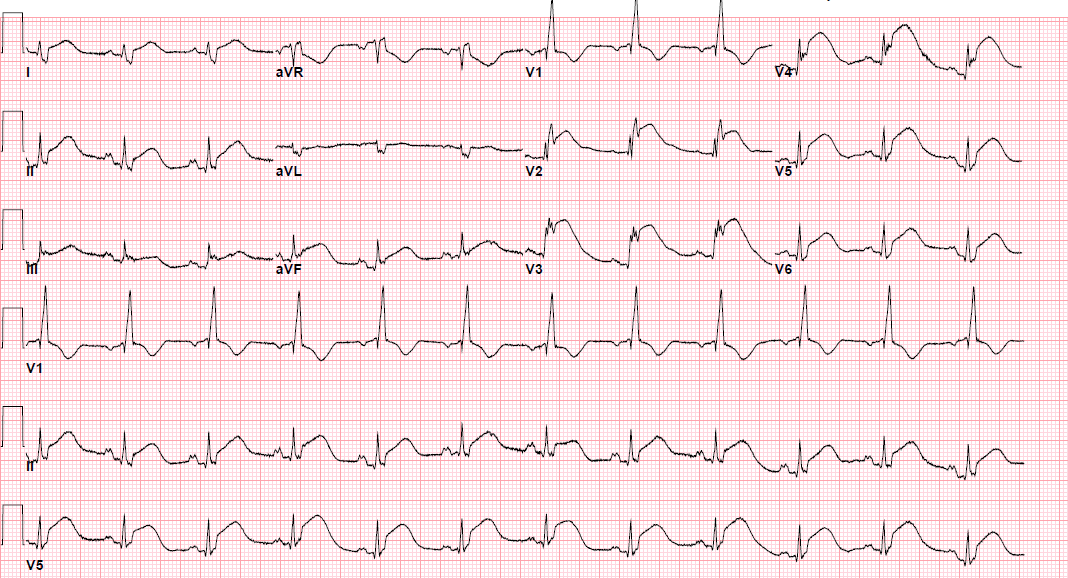
Post-procedure EKG
Case Overview
- Underwent complex intervention of the LM.
- Rotational atherectomy was done leading to a minor dissection which evolved into a Type F dissection with AVC after pre-dilatation.
- Patient became hemodynamically unstable with VF arrest that was successfully treated with a single defibrillation.
- Two stents were placed in the LM extending into the LAD with restoration of flow.
- An additional stent was placed, effectively treating the dissection.
- Troponin-I peaked at 43.8 ng/mL and CK-MB peaked at 108.8 ng/mL.
- Patient was discharged home 7 days after without further sequelae.
Learning Objectives
- What is the likely explanation or reason why the complication occurred?
- Wire bias altering the trajectory of the rotational atherectomy device.
- How could the complication have been prevented?
- We should have performed rotational atherectomy starting with a smaller 1.25mm rota burr and upsized to a larger 1.5mm or 1.75mm rotational burr. This approach would have better modified the plaque in the tortuous distal LM and in turn reduce wire bias which was responsible for altering the trajectory of the rotational burr while performing rotational atherectomy.
- Could have considered using an alternative atherotomy device such as a cutting balloon.
- Use of a guide catheter which provides coaxial engagement will help reduce unfavorable guidewire bias.
- Is there an alternate strategy that could have been used to manage the complication?
- Sometimes wire bias can possibly be prevented by use of a Rota Extra Support wire instead of a Rota Floppy wire. A stiffer wire can be used to straighten the vessel or lesion to lessen the resistance and possibly reduce wire bias.
- What are the important learning points?
- “Failure to plan is planning to fail” – Benjamin Franklin
- Need to be extremely cautious when using atherectomy devices in extremely tortuous vessels. Wire bias can occur in tortuous vessels, increasing the risk of dissection or perforation.
- Minor dissections are expected after atherectomy or balloon dilatation of a calcified vessel. Rarely, do they result in Type D to F dissections. Therefore, it is advised to keep appropriate balloons and stents ready when performing complex interventions to manage unexpected complications.
- Once a dissection is identified, rotational atherectomy should be stopped, and primary focus should be on maintaining wire position. Once assures the wire is in the true lumen, the dissection should be treated accordingly with balloon inflation and placement of a stent.
- Guidewire bias is when you have divergence from the central axis of the vessel and can result in ablation of normal tissue if the tension on the wall exceeds the elasticity of the vessel. To reduce sidewall tension:
- Keep the tip of the guidewire just beyond the lesion.
- Use a stepped-burr approach starting with an undersized burr is preferred.
- When advancing the burr, do so at low speed to reduce there is less tension in the wire.
- There are two kinds wires that can be used when performing rotational atherectomy:
- Rota Floppy Wire: Used in most cases, and is more flexible with a longer taper and shorter spring tip compared with the Rota Extra Support wire. This wire causes less vessel straightening, less wire bias, and allows for atherectomy along the greater curvature of an angulated lesion.
- Rota Extra Support Wire: Used in tortuous vessels or in situations when one suspects wire bias as a reason for the burr not crossing the lesion. Wire is stiffer, and with a shorter taper and longer spring tip compared with the Rota floppy wire. This wire causes the vessel to straighten out the tortuosity to allow the burr to advance to a lesion, and is useful in ablation of plaque at the lesser curvature of angulated lesions, aorto-ostial lesions and distal lesions. However, this wire can result in proximal vessel spasm and ‘pseudo-stenosis’.
- A stiffer guidewire does not always result in unfavorable bias, and may result in a favorable bias, especially when performing rotabalation of an angulated and heavily calcified lesion.