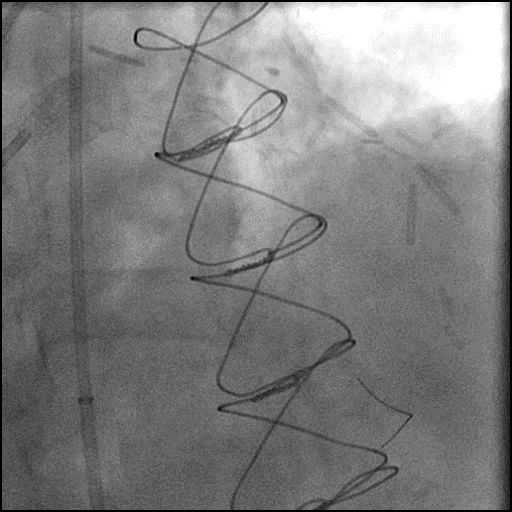
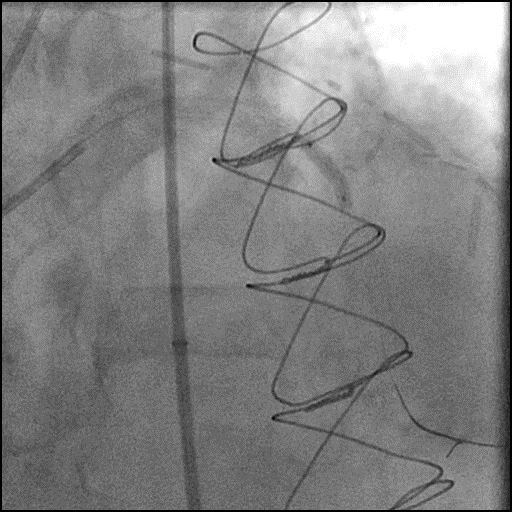
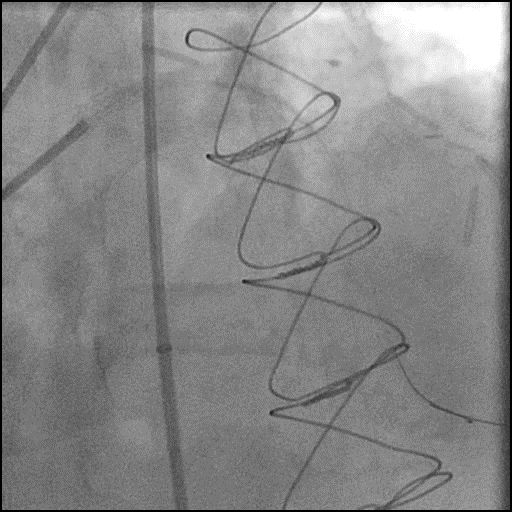
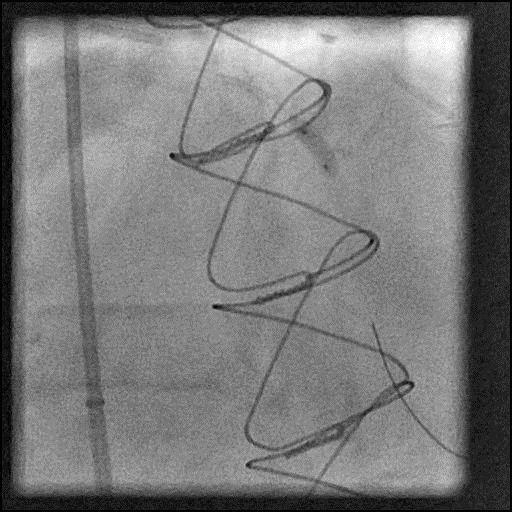
ABRUPT VESSEL CLOSURE (AVC) - mobile
- AVC is the commonest major complication of PCI1
- Incidence: 0.3% [used to be 3% in pre-stent era]2
- Risk factors:3
- Proximal vessel tortuosity
- Diffuse lesion
- Pre-existing thrombus
- Degenerated vein graft
- Extremely angulated lesion
- Unstable angina
- Multivessel disease
- Female gender
- Chronic renal failure
- Common causes:3
- Coronary dissection
- Intracoronary thrombus formation
- Native thrombus (or atheroma) embolization
- Air injection
- Coronary no-reflow
- Coronary vasospasm
- Classification of coronary perforation: As per the National Heart, Lung and Blood Institute scheme, types A–F classification remains useful to describe the severity of luminal injury:4
| Type A | Minor radiolucency within the coronary lumen without dye persistence |
| Type B | Parallel tracks or double lumen separated by a radiolucent area during angiography without dye persistence |
| Type C | Extraluminal, persisting extravasation of contrast |
| Type D | Spiral luminal filling defects |
| Type E | Persistent lumen defect with delayed antegrade flow |
| Type F | Filling defect accompanied by total coronary occlusion |
- Prevention:
- Maintain ACT > 300 throughout procedure
- Make sure interface is free of air
- Avoid high-pressure balloon dilatation or stenting
- Avoid unnecessary post-dilatation and very long stents
- Use distal protection devices in vein graft PCI
- Be careful when retrieving delivery system after stent implantation
- Avoid geographical miss during stenting
- Avoid aggressive post-dilatation at the stent edges
- Be careful while positioning wire distal tip in tortuous vessel
- Management: Abrupt closure results in acute ischemia manifesting as ECG changes, hypotension, bradycardia, chest pain and ventricular arrhythmias. The first step is to identify the underlying cause of AVC and then treat it accordingly.
- Immediate priority should be to ensure intraluminal position of coronary guidewire and, if in doubt, an over-the-wire balloon catheter or Twin-Pass or other microcatheter should be advanced distal into the target vessel to allow minimal contrast media injection and confirm wire position.
- If intraluminal guidewire position is confirmed, the most likely mechanism underlying AVC is dissection or intraluminal thrombus. Prompt balloon inflation should be attempted to establish antegrade flow. If flow returns immediately after balloon inflation the likely cause of AVC is dissection and urgent stenting is useful for stabilizing.
- If the distal flow after balloon inflation is sluggish (TIMI 0 or 1), the likely cause of AVC is distal thromboembolism. Using a Twin-Pass or microcatheter to administer distal vasodilators can help reestablish flow.
- If initial contrast agent injection reveals guidewire position within a false lumen, careful exploration of the occluded segment using a second guidewire must be performed.
- Aspiration thrombectomy and Glycoprotein IIb/IIIa antagonists may be helpful if acute closure is due to thrombus. Control of anticoagulation is of paramount importance to avoid thrombotic occlusion of stented artery. ACT should be measured every ~30 minutes to keep ACT > 300 throughout the procedure and to ensure dose of anticoagulation is adjusted accordingly. If ACT is not reaching therapeutic levels consider resistance to anticoagulant and a possible reason for suspected thrombus formation causing AVC.
- Intravenous fluids, vasopressors, inotropes and intra-aortic balloon pump (IABP) should be considered for unstable hemodynamics.
- Emergency CABG should be considered if patient has persistent AVC depending on the location of the occlusion, patient’s clinical condition and assessment of risks and benefits.
Coronary slow flow/no-reflow phenomenon
Slow flow/No-reflow is an acute reduction in coronary flow (TIMI grade 0–1) in a patent vessel with absence of dissection, thrombus, spasm, or high-grade residual stenosis at the original target lesion.5 The underlying mechanism is complex and not completely understood, but some proposed mechanisms include distal embolization of calcium, plaque or thrombus and microvascular spasm caused by release of vasoconstrictor substances like serotonin and thromboxane, oxidative stress, and reperfusion injury.6 Clinical and lesion characteristics associated with higher incidence of no-reflow include left ventricular systolic dysfunction or hemodynamic instability, long calcified lesions, ostial lesions, chronic total occlusion of right coronary artery, thrombotic lesions, and vein graft lesions. Use of rotational atherectomy is also associated with a higher incidence of no-reflow.- Prevention
- Direct stenting whenever feasible
- Use of distal embolic protection devices for vein graft interventions.
- Aspiration thrombectomy in STEMI cases if there is large thrombus burden
- For cases involving rotational atherectomy, the use of rota flush, small initial burr sizes, shorter rotablation runs, avoiding drops in rotations per minute (RPMs), and prevention of hypotension/bradycardia
- Management: Coronary no re-flow must be immediately differentiated from AVC due to dissection as placement of stent in a vessel with no reflow may worsen the situation. Exclusion of dissection, thrombus, spasm, or high-grade residual stenosis at the original target lesion suggests no-reflow.
- Stabilize hemodynamics with medications/intra-aortic balloon pump (IABP)
- IC verapamil (100–200 μg)7
- IV adenosine (10–20 μg)
- IC nitroprusside (50–200 μg)
- Moderately forceful injection of blood or saline through the manifold
- GPIIb/IIIa agents, IV cangrelor may also be helpful
Air Embolism
Intracoronary air embolism is a potentially lethal but rare complication. It could result in hypotension, hemodynamic collapse, cardiac arrest, and in rare cases death. Coronary air embolism is almost always iatrogenic. It occurs most commonly:- When catheters are not adequately aspirated and flushed
- During introduction or withdrawal of a guidewire, balloon catheter, or other interventional devices
- As a result of rupture of a balloon during high pressure inflation
- During intracoronary medication injection
- Diagnosis: Coronary air embolism is detected fluoroscopically as intracoronary filling defects during dye injection. It could also be seen as abrupt cutoff of a vessel secondary to occlusion of distal circulation with air column. Clinically small air embolism may be asymptomatic. Larger air embolism may present as chest pain, hypotension, ischemic EKG changes, or cardiac arrest.
- Prevention
- Do not engage the left main coronary when pulling out the guidewire unless the patient has excessive aortic tortuosity or an enlarged aortic root.
- Do not connect the manifold to the catheter with the flush running. This may lead to an air embolism if the catheter already has a column of air inside it.
- Draw back at least 2 cc of blood into the injection syringe and make sure that the interface is free of air prior to injection.
- Inject some dye into the ascending aorta prior to engaging left main.
- Always ensure that all the catheters and tubings are aspirated, flushed and free of air.
- Take adequate care when prepping stents or balloons and ensure that the syringe tip is facing downwards.
- Always inject with the syringe tip facing downwards
- Treatment
- Put patient on 100% oxygen.
- Flush air free saline vigorously into the coronary arteries. Aspirate blood and air column via guide catheter and reinject saline forcefully back into coronary arteries.
- Administer IV phenylephrine 200 μg for hypotension. Repeat, as needed every minute. If significant hypotension or hemodynamic collapse is present, push IV 1 cc epinephrine (1:10,000 dilution).
- Intracoronary injection of vasodilators (adenosine, nitroprusside, verapamil) may be attempted.
- Supportive measures should be instituted (IABP for persistent hypotention) and patient admitted to intensive coronary care unit for further monitoring
Coronary Vasospasm
Coronary vasospasm can be induced by PCI secondary to endothelial denudation and nitric oxide loss.- Some cases are catheter-induced which is caused by a contact of a catheter without balloon deployment. It is usually short-lived and is most prone to occur at the ostium of the right coronary artery (RCA). The left main is less susceptible to ostial spasm
- Rotablator cases are more prone to vasospasm
- Diagnosis
- Coronary vasospasm is detected by presence of EKG changes of ST segment elevation in association with angina, and then EKG completely returns to baseline upon resolution of symptoms.
- The definitive diagnosis of coronary vasospasm is made angiographically by demonstration of reduction of luminal diameter in a discrete segment of the vessel, which is proven reversible by the administration of intracoronary vasodilators.
- Treatment
- Initial step is intracoronary vasodilatation with IC calcium channel blockers and/or nitrates [nitroglycerin 100-300 mcg, verapamil 100 mcg/min, up to 1.0-1.5 mg, nicardipine 100-300 mcg, nitroprusside 100-300 mcg]
- IV atropine can be useful if there is associated hypotension of bradycardia
- If vasospasm persists, remove all hardware and leave the guidewire in place to maintain position. This may minimize distal vessel spasm
- Repeat prolonged PTCA for 2-5 minutes at low pressures (1-4 atmospheres)
- Stenting should be reserved in cases if all the above measures have failed, as it may lead to propagation of spasm to a new location
- Refractory vasospasm may be indicative of dissection, which is also an indication for stenting
Abrupt Vessel Closure Summary
- Dissection
- Minor dissections - usually heal without clinical sequelae, no treatment required
- Major dissections - repeated prolonged low-pressure balloon [distal vessel], stenting [Proximal/mid vessel segment or impaired flow due to dissection]
- Thromboembolism
- Twin-Pass or microcatheter to administer vasodilators distally
- Check ACT to keep > 300. Consider starting IV Cangrelor or bailout GPIs
- Balloon dilatation and/or thrombus aspiration in case of stent thrombosis
- Stenting in case of thrombosis in unstented vessel segment
- No-reflow
- Intracoronary Adenosine, Nitroprusside, Nicardipine, Verapamil, or GPI’s
- A transit catheter or over-the-wire balloon should be used to deliver the vasodilators to the distal microvasculature
- Insertion of IABP to improve flow
- Air embolism
- Start 100% oxygen
- Flush air free saline vigorously into the coronary arteries. Aspirate blood via guide catheter and reinject forcefully back into coronaries
- IV phenyl epinephrine or epinephrine as needed
- Intracoronary injection of vasodilators
- Vasospasm
- Intracoronary Nitroglycerin, Adenosine, Nitroprusside, Nicardipine, or Verapamil
- IV fluid bolus and/or atropine as needed
- Remove all hardware and leave the guidewire in place to maintain position
- Repeat prolonged PTCA for 2-5 minutes at low pressures (1-4 atmospheres)
- Unknown etiology
- Maintain wire position distally and pass a microcatheter distally to inject contrast
- If flow distally, problem at site of vessel closure and needs to be investigated
- If no flow distally, consider no reflow and give IC vasodilators
References
- de Feyter P.J., de Jaegere P.P.T., Murphy E.S., Serruys P.W. (1992) Abrupt coronary artery occlusion during percutaneous transluminal coronary angioplasty. Am Heart J 123:1633–1642.
- Francesco Giannini, Luciano Candilio, Satoru Mitomo, Neil Ruparelia, Alaide Chieffo, Luca Baldetti, Francesco Ponticelli, Azeem Latib, Antonio Colombo. Practical Approach to the Management of Complications During Percutaneous Coronary Intervention. J Am Coll Cardiol Intv. 2018 Sep, 11 (18) 1797-1810.
- Klein L. (2005) Coronary complications of percutaneous coronary interventions: a practical approach to the management of abrupt closure. Catheter Cardiovasc Interv 64:395–401.
- Huber MS, Mooney LF, Madison J, et al. Use of a morphologic classification to predict clinical outcome after dissection from coronary angioplasty. Am J Cardiol 1991;68:467–71.
- Rezkalla S.H., Kloner R.A. (2002) No-reflow phenomenon. Circulation 105:656–662.
- Piana R., Paik G., Moscucci M., et al. (1994) Incidence and treatment of “no-reflow” after percutaneous coronary intervention. Circulation 89:2514–8.
- Su Q., Li L., Liu Y., et al. (2013) Short-term effect of verapamil on coronary no-reflow associated with percutaneous coronary intervention in patients with acute coronary syndrome: a systematic review and meta-analysis of randomized controlled trials. Clin Cardiol 36:E11–E16