Intravascular Brachytherapy
Mechanism: locally applied radiation can block or attenuate local tissue proliferation causing ISR.
Indication: to reduce the recurrence of in-stent restenosis (ISR).
Indication: to reduce the recurrence of in-stent restenosis (ISR).
Possible negative effects of brachytherapy:
- Delayed recurrent restenosis
- Late stent thrombosis because of prolonged inhibition of intimal formation
- Late vessel stent separation because of positive remodeling
- Geographical miss: reactive hyperproliferation at the edges because of less radiation dose
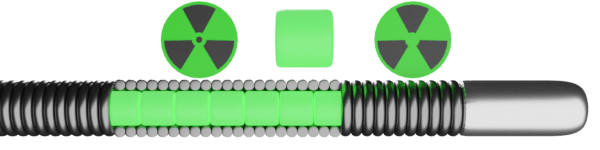
Types of brachytherapy: both gamma (low energy/high penetration) using 192-Ir and beta sources (high energy/low penetration) are effective, but only the beta source is clinically available
FDA-approved brachytherapy systems:
- Gamma: Cordis Checkmate (192-Ir)™ (not commercially available)
- Beta: Guidant Galileo (32-P)™ (not commercially available) and (Novoste Beta-Cath (90-Sr-Y)™
Novoste Beta-Cath™
- Guide selection: 6 F
- Wire: 0.014″
- Distal 1 cm rapid exchange length
- Selection of device and steps:
- Appropriate source train length (30 mm, 40 mm, or 60 mm) is selected based on the injury length and desired margin
- Balloon predilation of ISR is done
- No stent should be placed when IVBT is planned because of an increased risk of stent thrombosis
- Dual antiplatelet is continued for at least 3 years
- For a long lesion, serial delivery of catheters with a 1 mm overlap is recommended. We recommend adequate anticoagulation with bivalirudin (to an ACT of >300) and an additional dose of heparin IV just before delivery of the radiation dose.