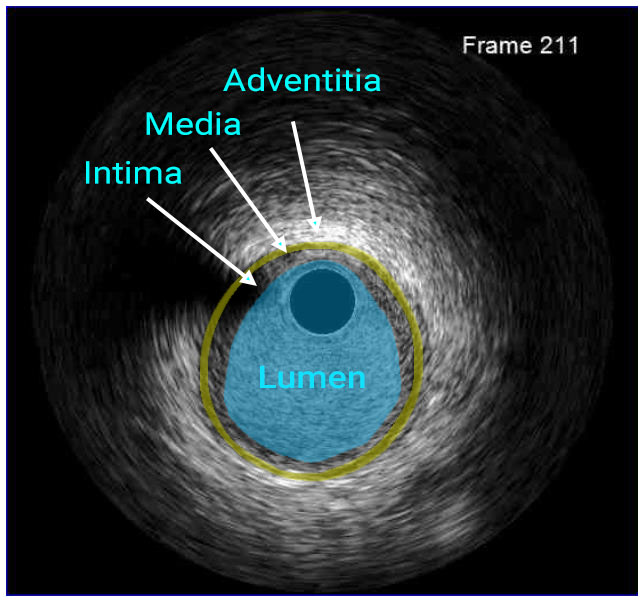
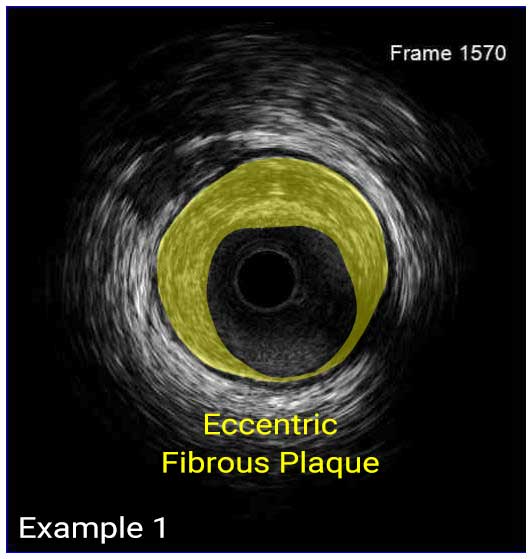
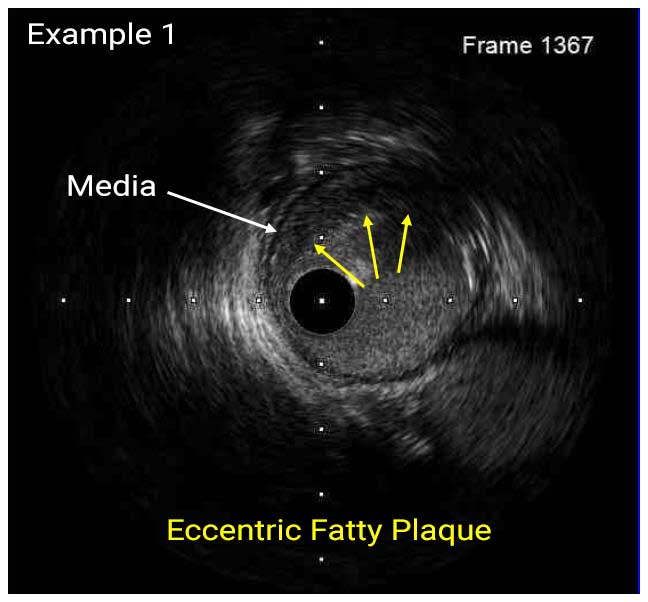
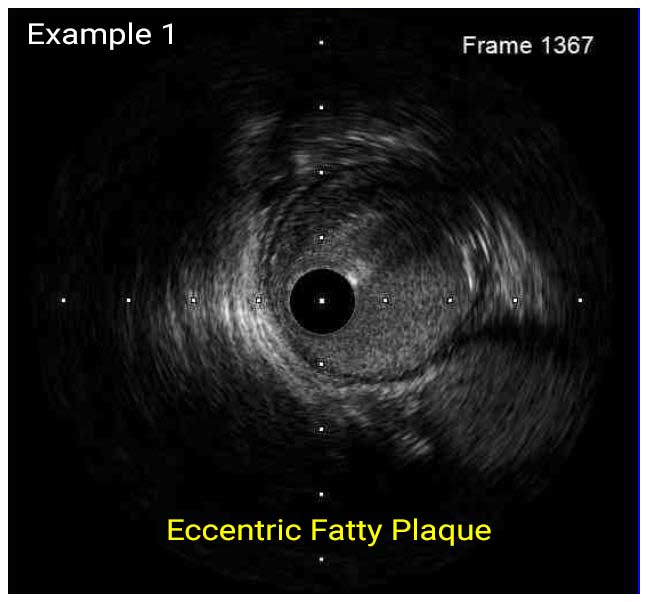
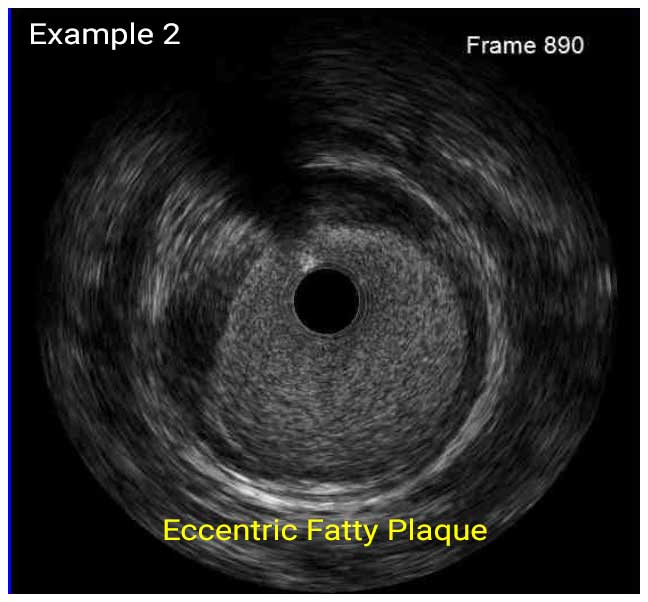
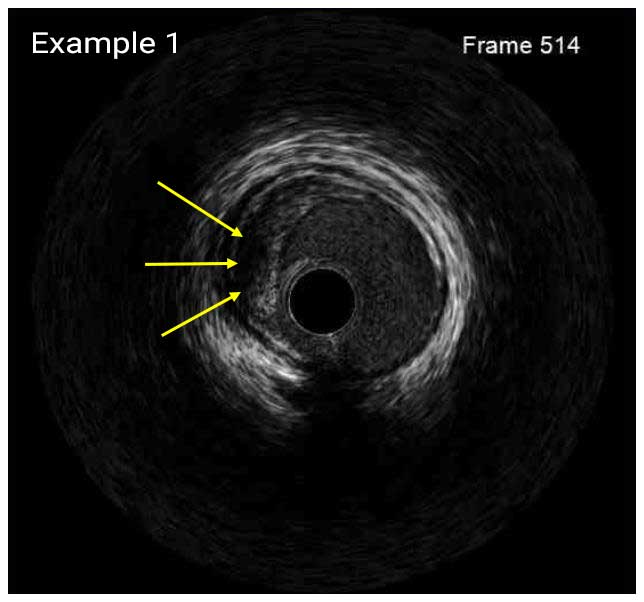
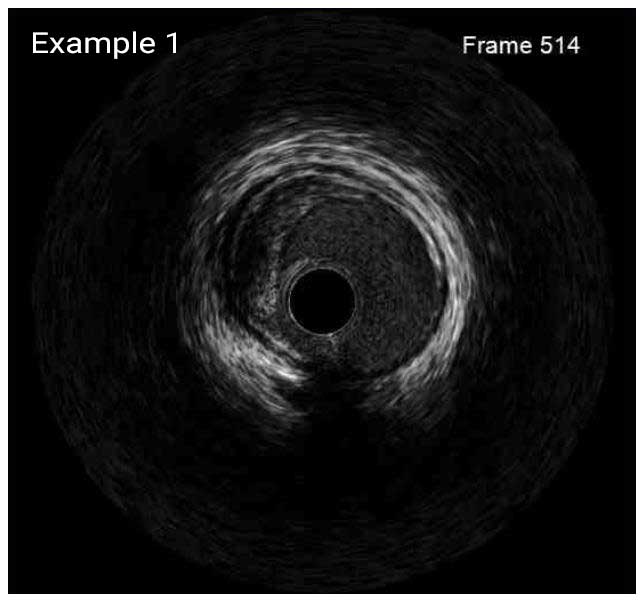
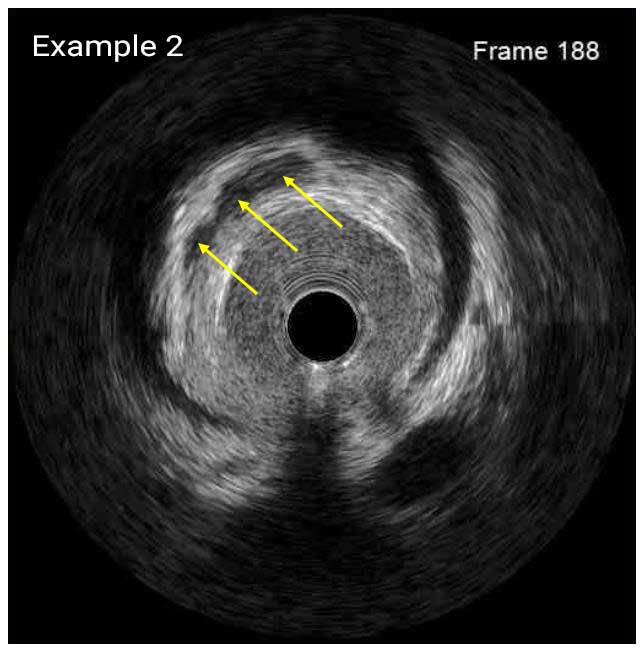
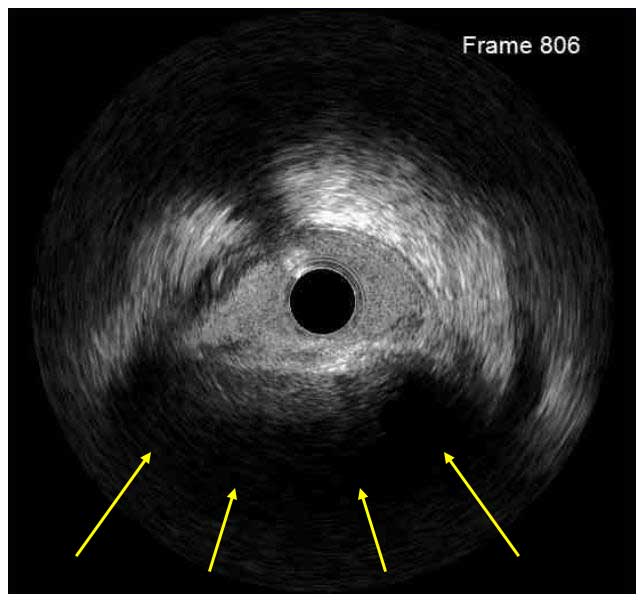
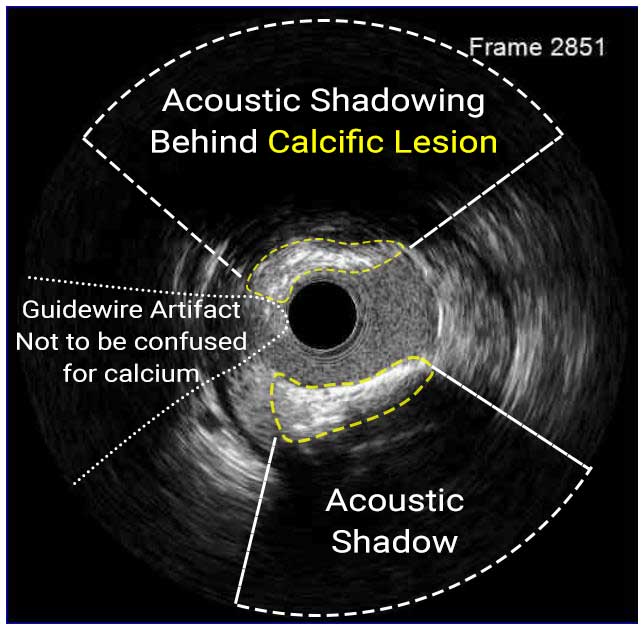
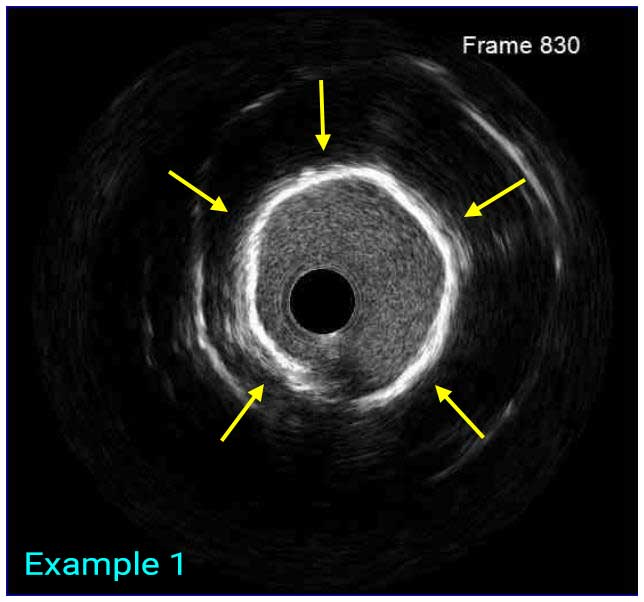
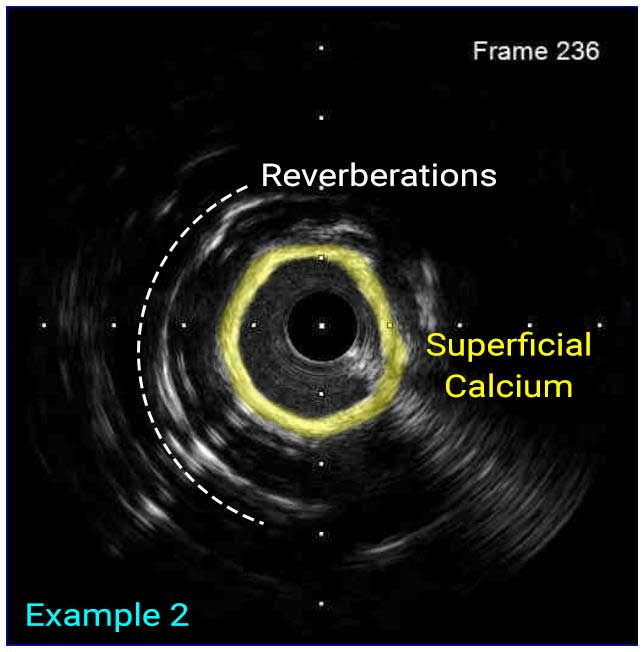
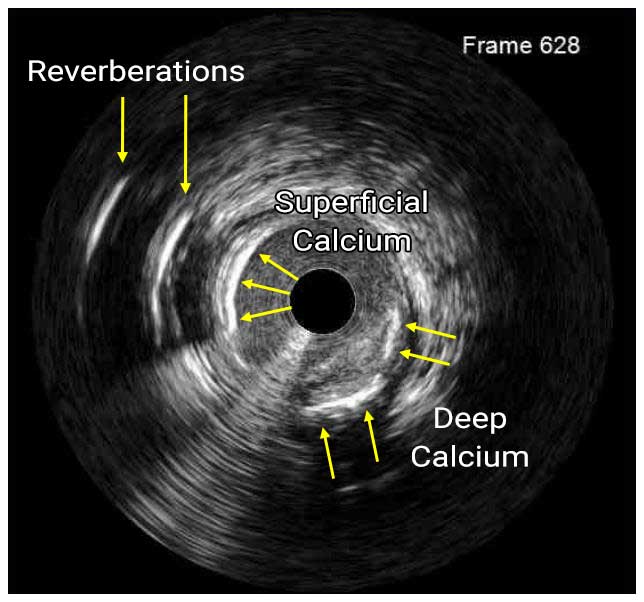
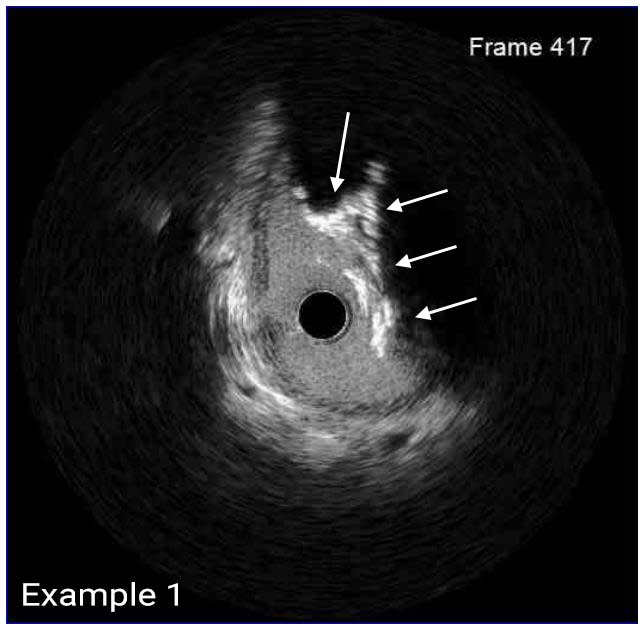
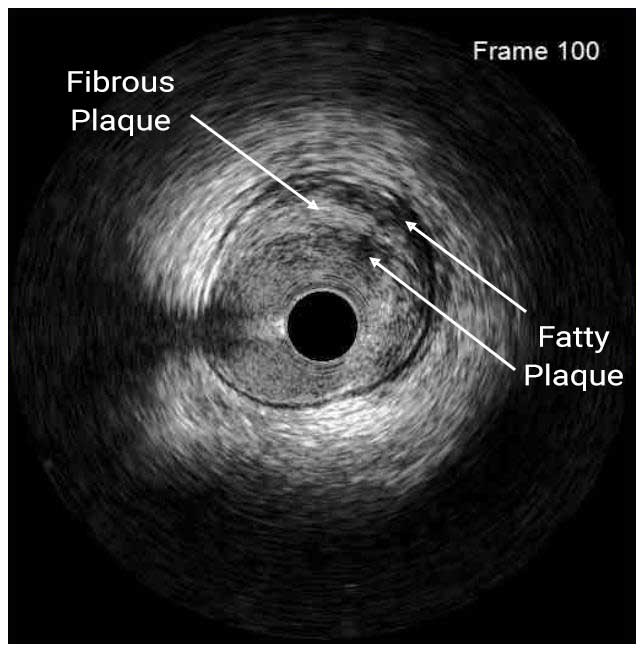
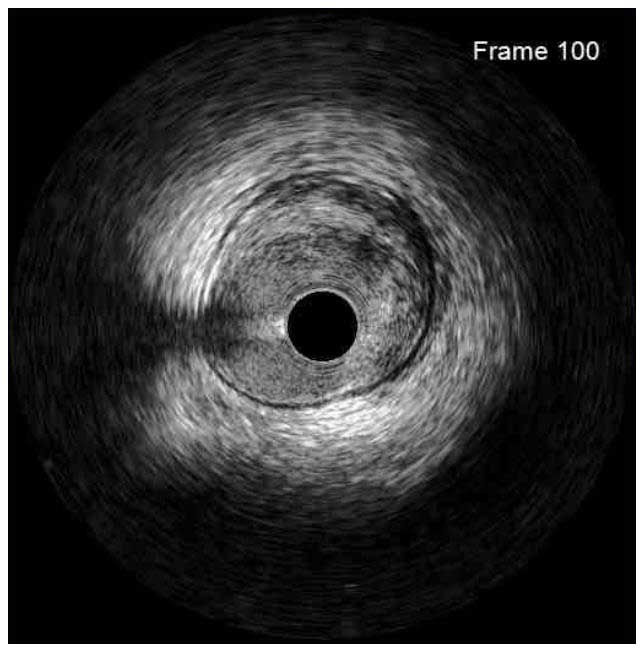
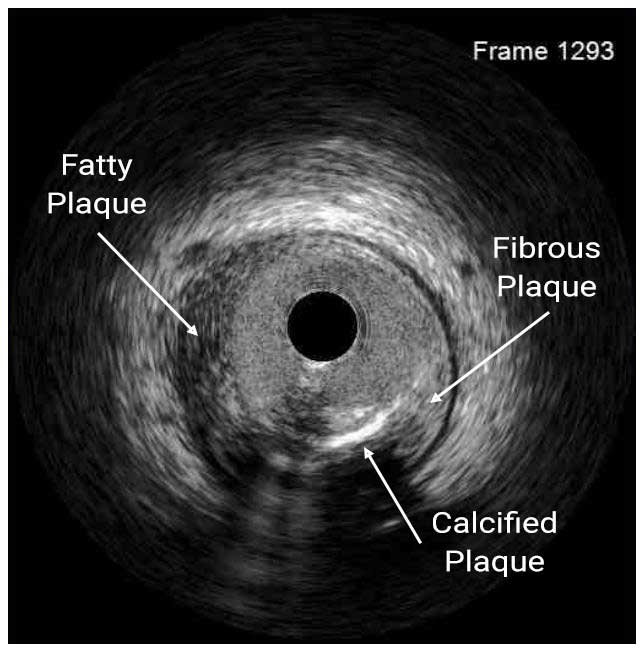
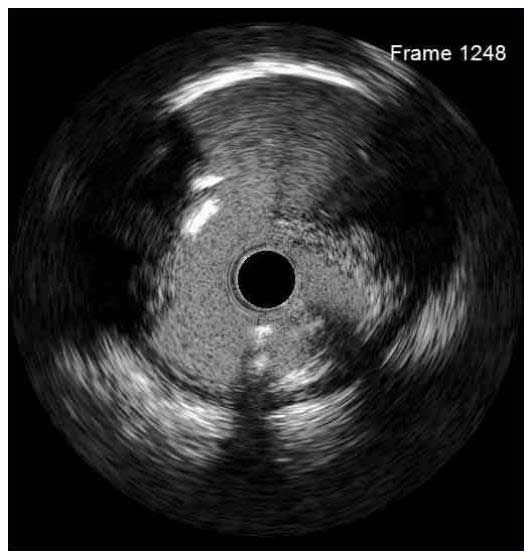
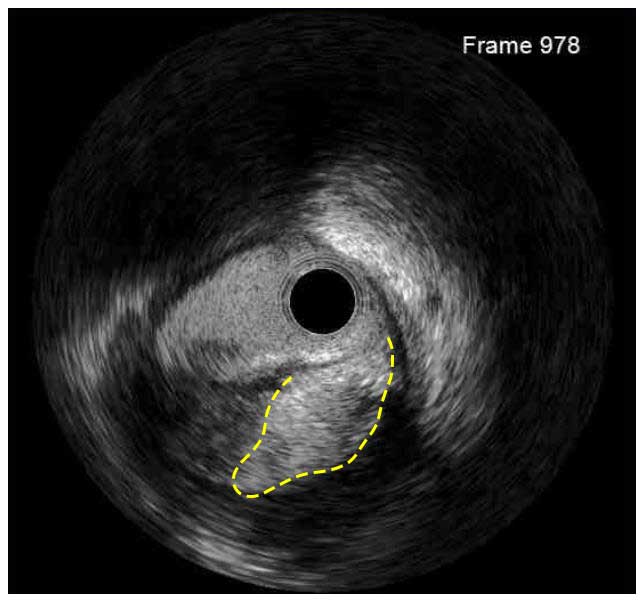
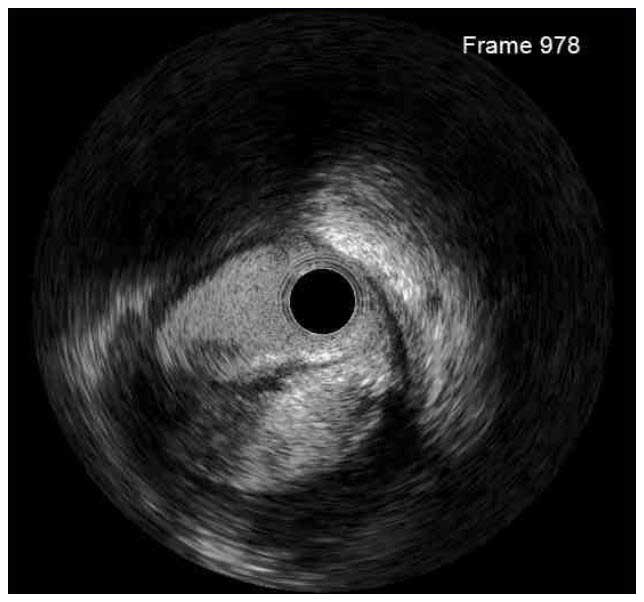
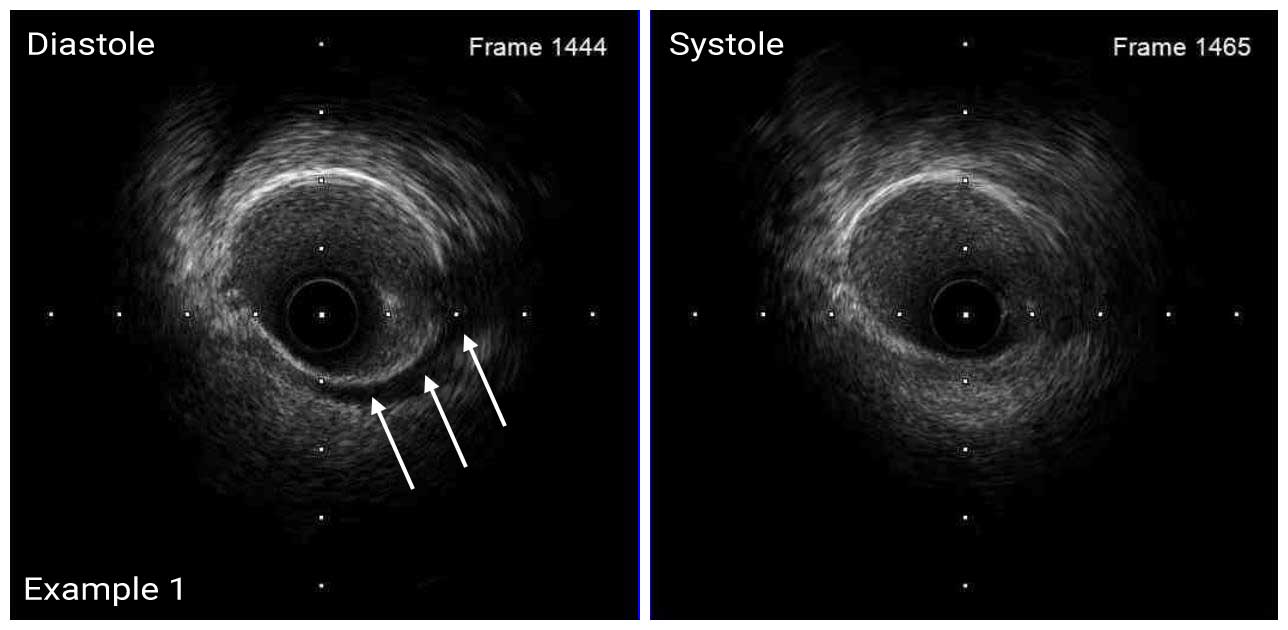
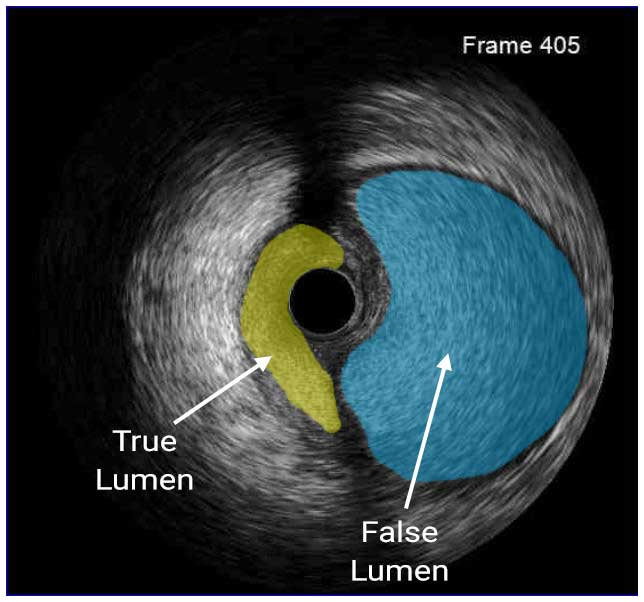
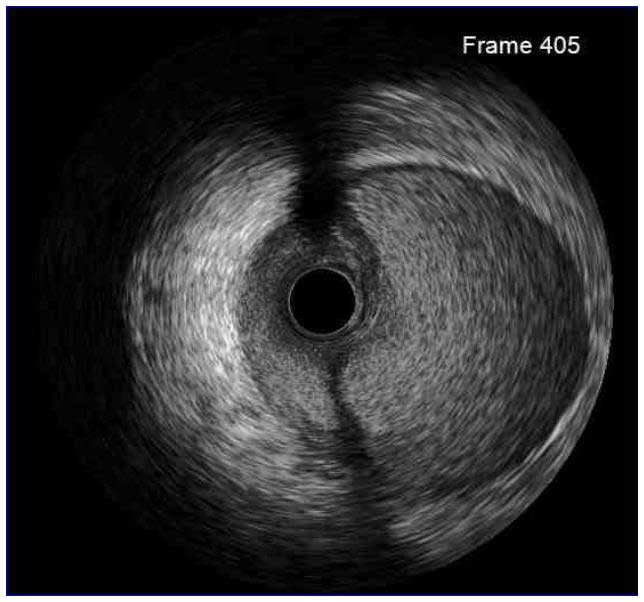
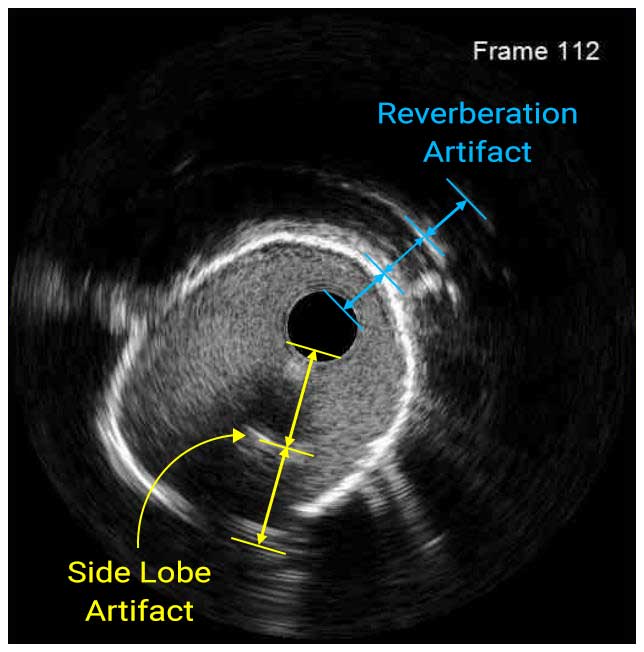
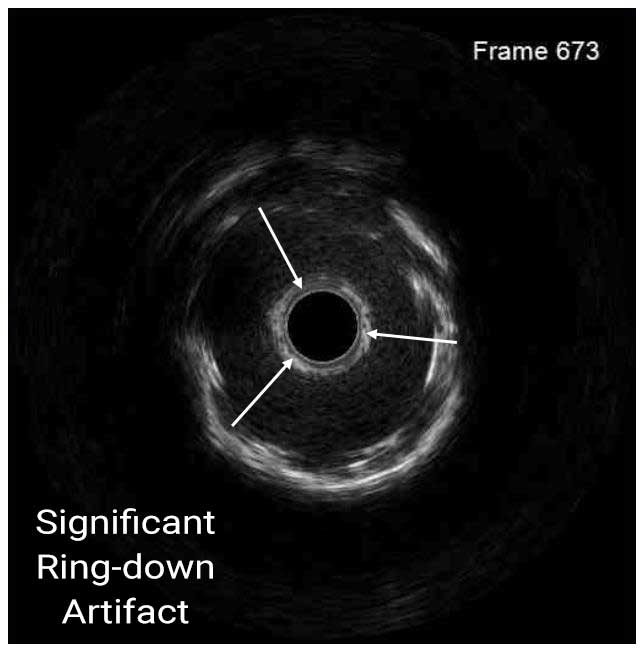
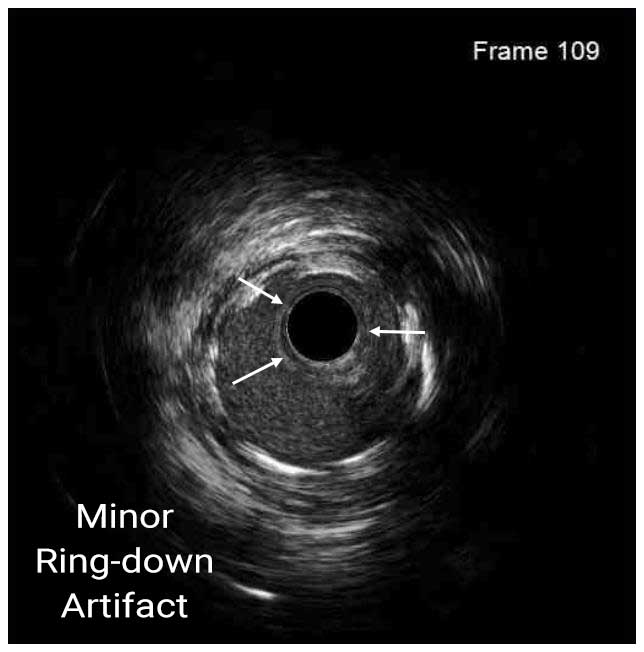
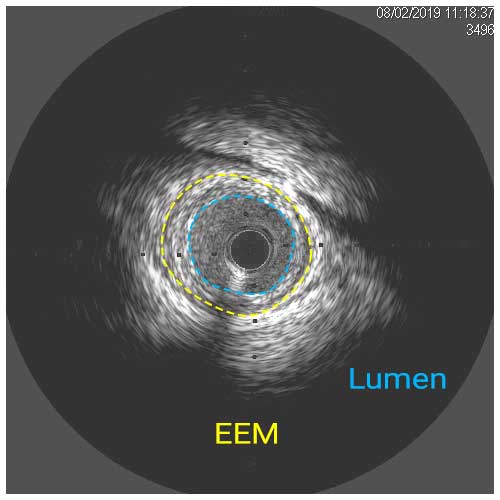
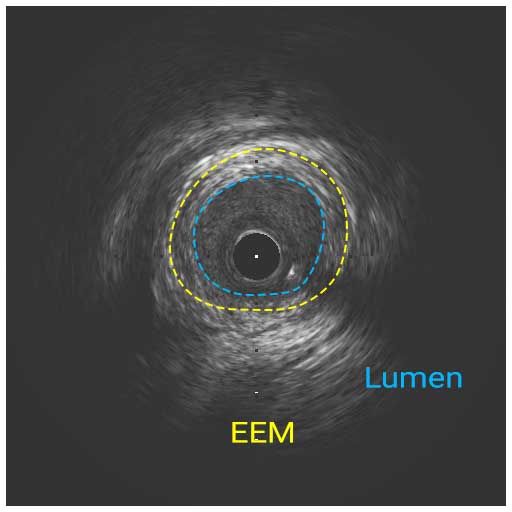
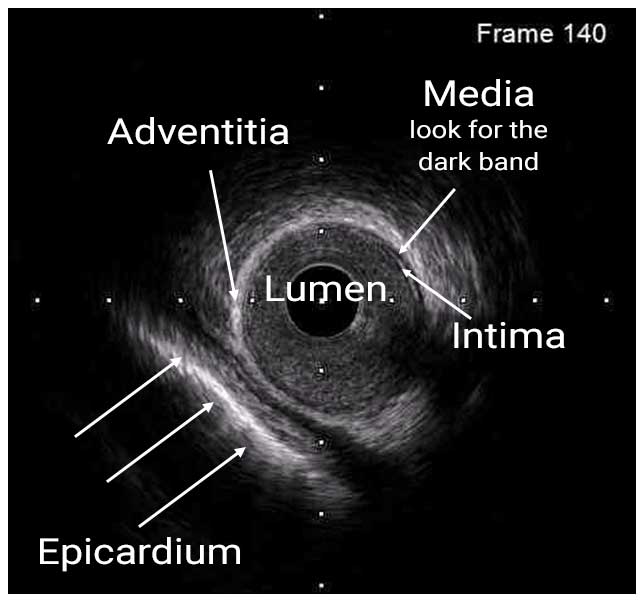
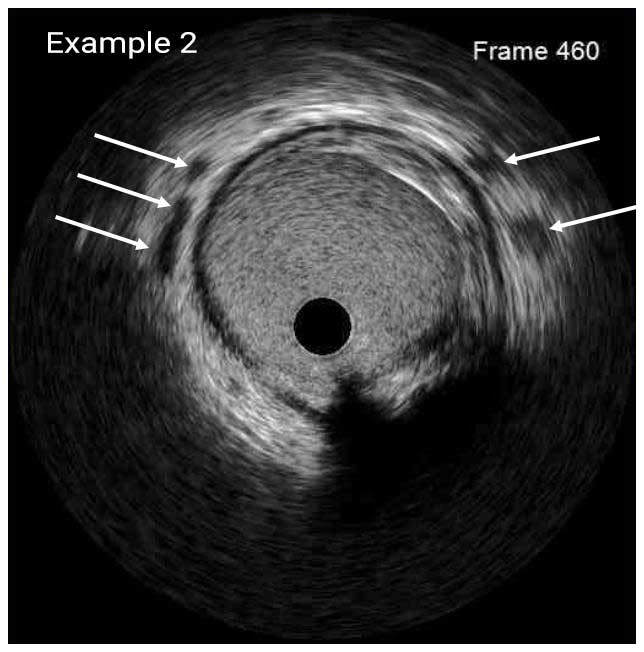
In a normal coronary artery, an ultrasound reflection is generated at two tissue interfaces: at the border between blood and the leading edge of intima and at the external elastic membrane (EEM) located at the media-adventitia border. The resulting three-layered structure consists of the tunica intima (bright, relatively echogenic layer compared to lumen and media), media (dark, less echogenic layer compared to intima) and adventitia (bright). Tunica media has lower ultrasound reflectance due to lower content of collagen and elastin (highly reflective materials) compared to intima and adventitia. The trailing edge of the intima, internal elastic membrane (IEM), cannot always be distinguished clearly on IVUS images. Similarly, IVUS cannot detect the outer border of adventitia due to comparable echoreflectivity of adventitia and periadventitial tissue.1,2
1. J Am Coll Cardiol. 2001 April, 37(5): 1478-92
2. Circulation. 2001;103:604-616
1. J Am Coll Cardiol. 2001 April, 37(5): 1478-92
1. J Am Coll Cardiol. 2001 April, 37(5): 1478-92
1. J Am Coll Cardiol. 2009, 2(1): 65-72
2. JACC Cardiovascular Interv. 2011, 4(5): 495-502
1. Circulation. 1995, 91(7): 1959-1965.
2. J Am Coll Cardiol. 2001 April, 37(5): 1478-92
1. Am J Cardiol 2011;108:1547–1551
1. J Am Coll Cardiol. 2001 April, 37(5): 1478-92
1. J Am Coll Cardiol. 2001 April, 37(5): 1478-92
2. Am J Cardiol. 2006 Jan 1;97(1):29-33
1. J Am Coll Cardiol. 2001 April, 37(5): 1478-92
2. Catheter Cardiovasc Interv. 2014 Dec 1;84(7):1115-22
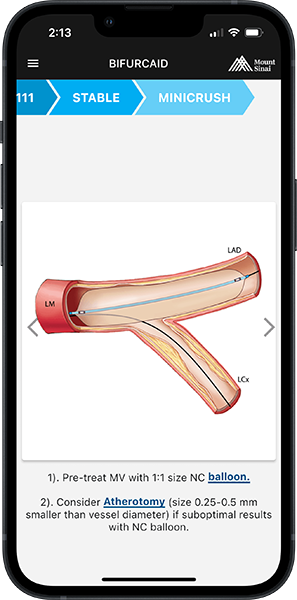
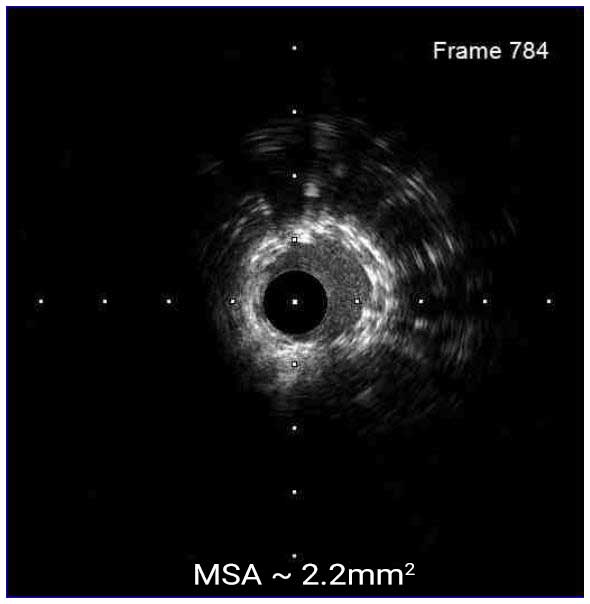
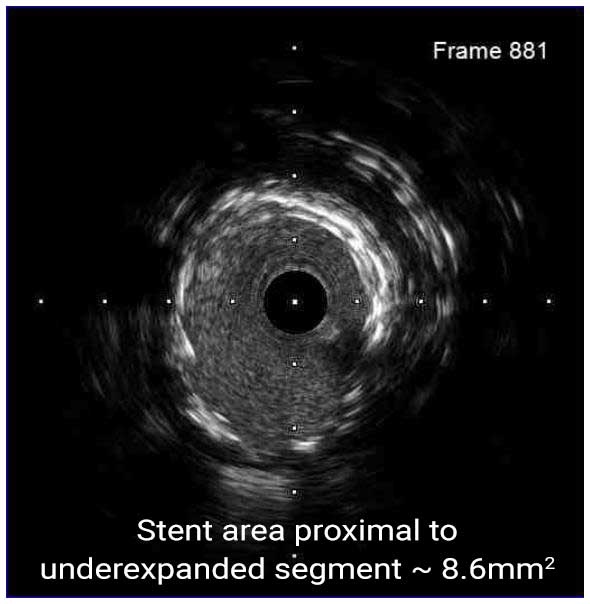
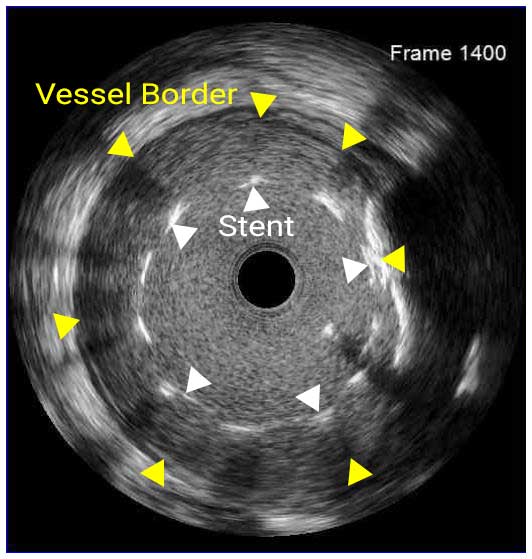
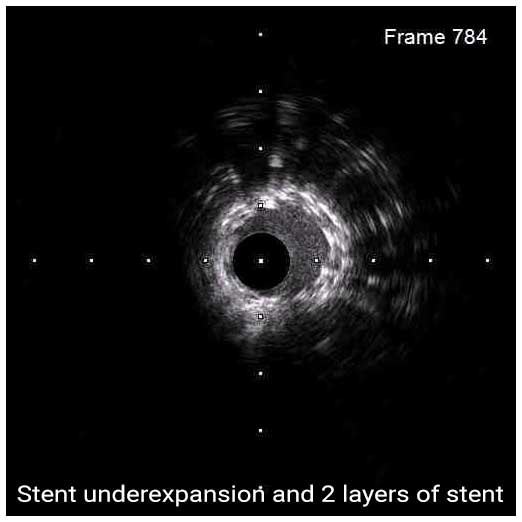
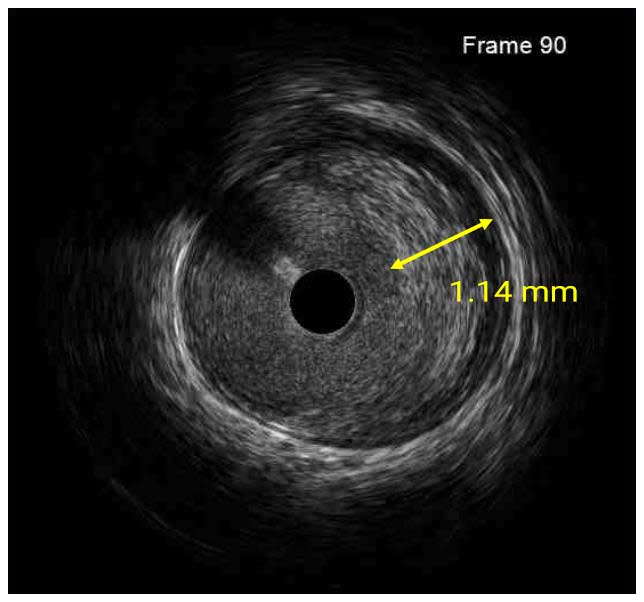
Stent underexpansion is the most consistent predictor of early stent thrombosis or restenosis. IVUS criteria for stent optimal expansion include relative stent expansion >90% (MSA divided by distal reference lumen area) or absolute stent expansion with the final MSA of >5.5 mm² in non-LM lesions, MSA >7 mm² distal LM, and MSA >8 mm² and proximal LM.1,2 The absolute cut-offs may not be achievable in small vessels, may result in stent undersizing in large vessels, and differ between BMS and DSE. In long stenoses, the lesion can be divided into proximal and distal halves, and the MSA within each half is compared to the nearby reference.
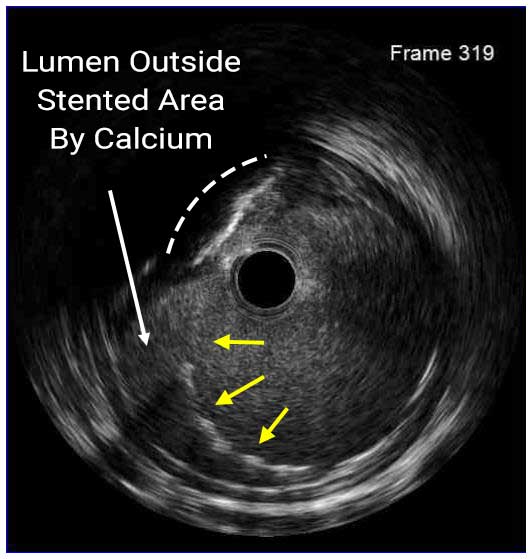
Stent underexpansion is frequently observed in heavily calcified coronary lesions, which might increase the risk of future adverse cardiac events. Therefore, lesion preparation with debulking devices or various plaque-modifying techniques is crucial prior to stent placement in these lesions.
1. JACC Cardiovascular imaging 2022;15:1799-1820
2. European heart journal 2018;39:3281-3300.
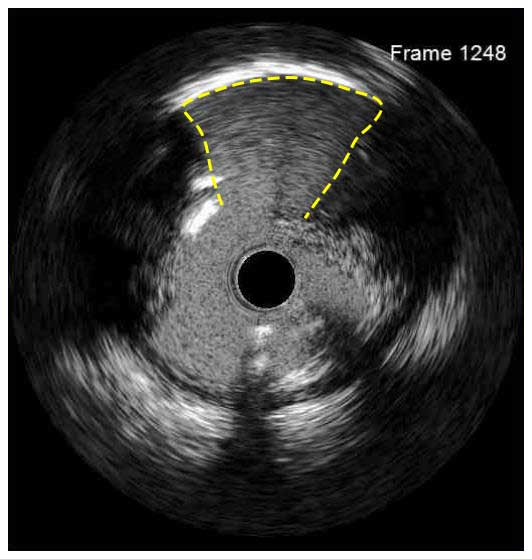
Stent malapposition is the lack of contact between the stent struts and the vessel wall. Stent malapposition and underexpansion can either occur independently or co-exist in the same segment. In contrast to stent underexpansion, there is no clear link between acute malapposition (in the absence of underexpansion) and future adverse events. Despite the current uncertainties regarding the clinical implications of malapposition, the findings from large stent thrombosis registries strongly suggest that large regions of malapposition should be avoided after stent implantation, especially at the proximal stent edge. Extensive malapposition within the proximal edge of a stent might lead to complications during subsequent PCIs such as guidewire getting behind stent struts (Case “Proximal LAD stent underexpansion and malapposition”).
1. JACC Cardiovascular imaging 2022;15:1799-1820
2. European heart journal 2018;39:3281-3300.
Stent struts are not in close contact with vessel wall and blood flow echo is observed between the stent strut and the vessel wall. In this case, after implantation of a 4.0 mm DES in the LMT, IVUS was performed and showed malapposition in the distal part of the stent. If it is not clear whether the patient has malapposition or not, a negative contrast method may be used , in which saline is injected into coronary artery to temporarily eliminate blood cells and ensure a good field of view.
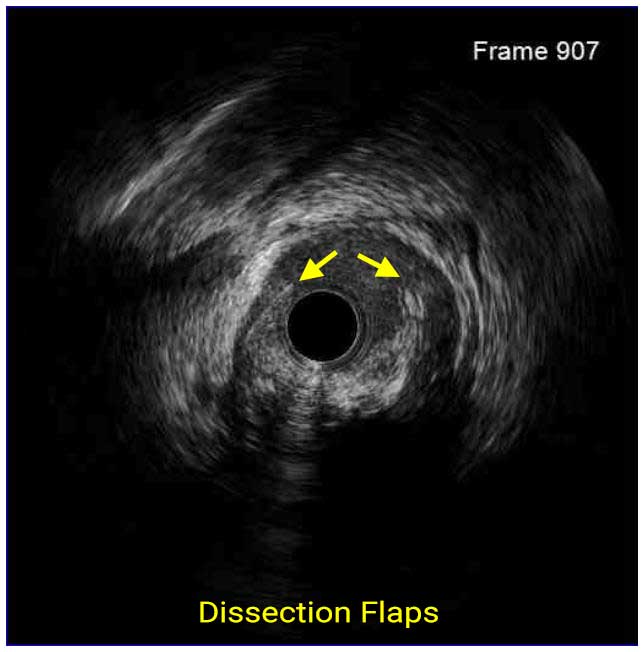
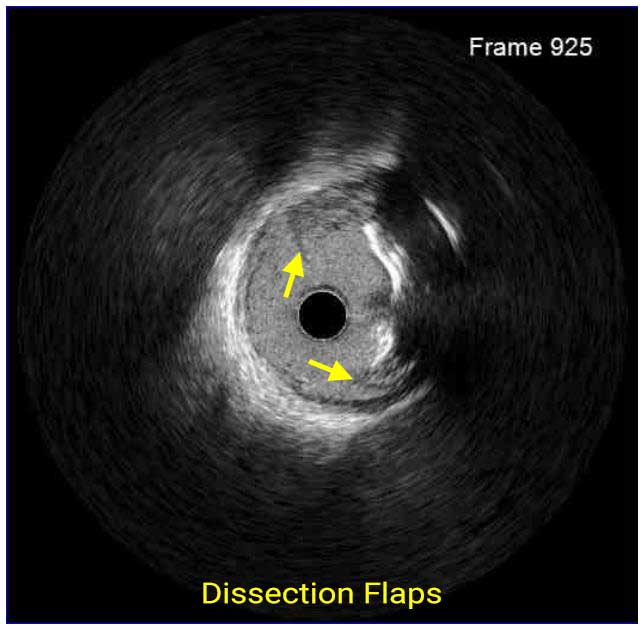
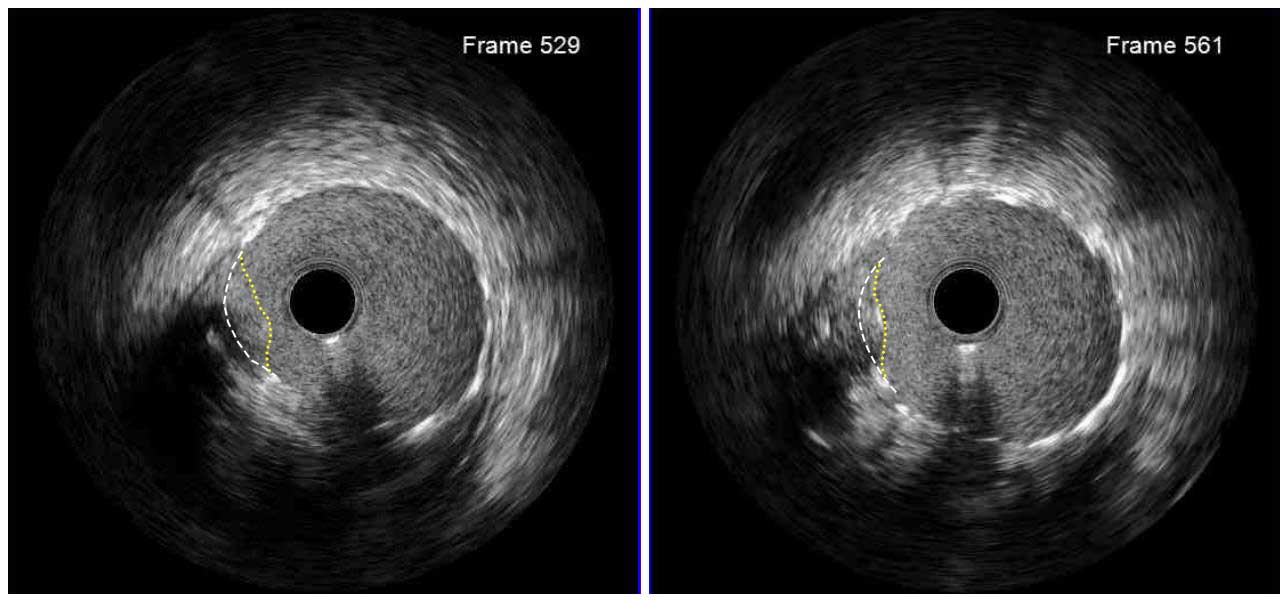
Following stent underexpansion, edge dissection after stent implantation is the second most important predictor of future adverse events. Dissections are classified by IVUS as intimal (limited to intima), medial (extending into the media), adventitial (propagating through EEM), intramural hematoma with blood accumulation within the media, and intra-stent separation of neointimal hyperplasia from stent struts.¹
While minor intimal dissections are unlikely to be clinically significant and do not require any additional treatment, extensive dissections disrupting the medial layer should be treated with additional stent implantation. Lateral extension >60°, longitudinal length >2 mm and involvement of medial or adventitial layers, especially at the distal stent edge, characterize the clinically significant dissections requiring correction.² Intramural hematomas represent another stent edge-related issue, which can increase the risk of future events. It might appear as edge stenosis on angiography, and the progression of uncovered hematoma may lead to early stent thrombosis.
1. J Am Coll Cardiol. 2001 April, 37(5): 1478-92
2. European heart journal 2018;39:3281-3300.
Plaque protrusion (prolapse) after stent implantation is commonly defined as tissue extrusion through the stent strut. Plaque protrusion is more likely to have adverse outcomes in the setting of ACS compared to stable CAD due to the differences in the composition of the protruding tissue. PCI should be considered in the setting of ACS or when the effective lumen area is significantly reduced. Stenting itself cannot reduce plaque volume, therefore stent placement in fatty, high-capacity plaque can cause plaque compression and protrusion through the stent struts into the vessel lumen. IVUS can show plaque protruding into the stent with a slightly high echo luminance, however these plaques are often difficult to characterize due to their similarity in signal and brightness to thrombus.1,2
1. European heart journal 2018;39:3281-3300.
2. JACC Cardiovascular imaging 2022;15:1799-1820
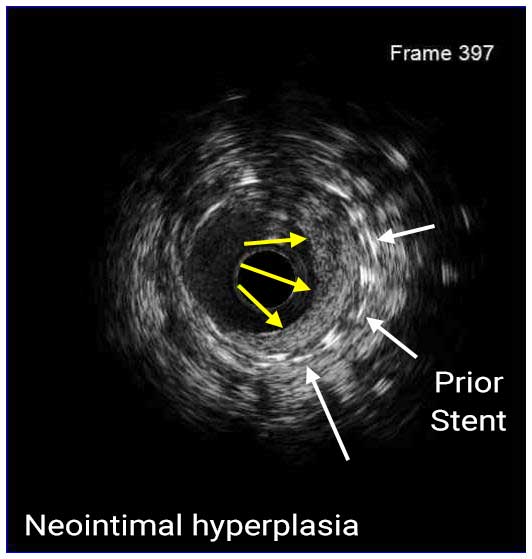
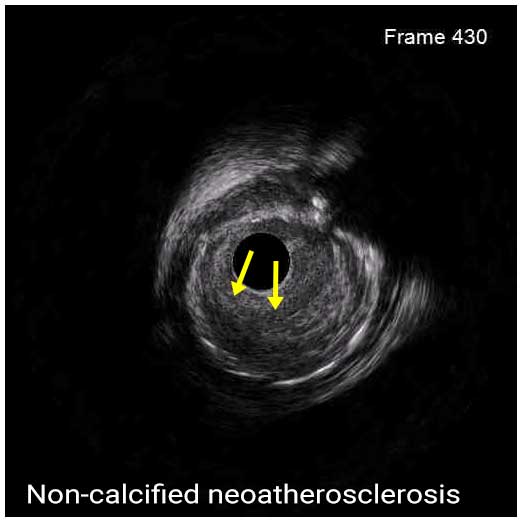
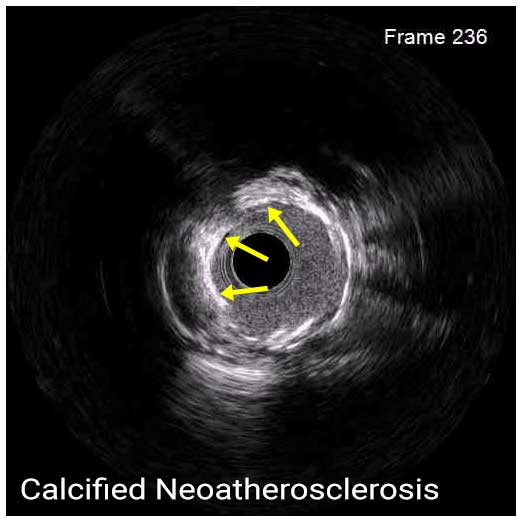
IVUS can help overcome inherent limitations of angiography in the assessment of ISR lesions and identify the underlying mechanism of the disease in order to guide the optimal treatment. Based on IVUS imaging, ISR can be classified as mechanical (stent underexpansion or rupture), biological (neointimal hyperplasia, non-calcified or calcified neoatherosclerosis), mechanical and biological combined, and multiple stent layers (>2). Neointimal hyperplasia, a common cause of ISR of DES, appears as a bright, homogeneous and uniform (in most cases) layer of tissue. Neoatherosclerosis is characterized by the presence of intrastent plaque with lipid or calcium echogenic characteristics. While non-calcified neoatherosclerosis demonstrates high attenuation, calcified neoatherosclerosis appears bright on IVUS.
1. Circ Cardiovasc Interv. 2019;12(8):2-8
2. Cardiov Revasc Medicine. 2021; 33:62-67
1. JACC Cardiovasc Imaging. 2017 Aug;10(8):869-879
Saphenous vein grafts (SVGs) after coronary artery bypass grafting (CABG) have different structure and morphology compared to native coronary arteries. Bypass grafts have no side branches and their walls do not originally have EEM, however, vein grafts undergo “arterialization” within the first weeks after implantation. The morphological changes include intimal fibrous thickening, medial hypertrophy, and lipid deposition (1). The early adaptive changes predispose to later atherosclerosis with occlusive plaque detectable in vein grafts within the first year. EEM is measured by tracing the outer border of the sonolucent zone. All other measurements including plaque plus media and plaque burden are calculated in a similar fashion to native coronary artery disease.¹
1. J Am Coll Cardiol. 2001 April, 37(5): 1478-92
Coronary disease is a major factor limiting long-term survival after cardiac transplantation representing the major cause of death after the first year post transplant. Cardiac allograft vasculopathy (CAV) is an accelerated form of coronary artery disease (CAD) characterized by concentric fibrous intimal hyperplasia along the length of coronary vessels. IVUS is an optimal tool for early detection and diagnosis of CAV. The most rapid rate of progression of intimal thickening occurs during the first year, followed by slow but inexorable progression over time. The presence of moderate to severe intimal thickening by IVUS is predictive of the future development of angiographically apparent CAV. Rapidly progressive CAV, defined as an increase of ≥0.5mm in maximal intimal thickness within the first year after heart transplantation, is associated with a significantly increased risk of all-cause death, myocardial infarction, and the subsequent development of angiographically severe CAV.1,2
1. J Am Coll Cardiol. 2001 April, 37(5): 1478-92
2. Circulation. 2008 Apr 22;117(16):2131-41.
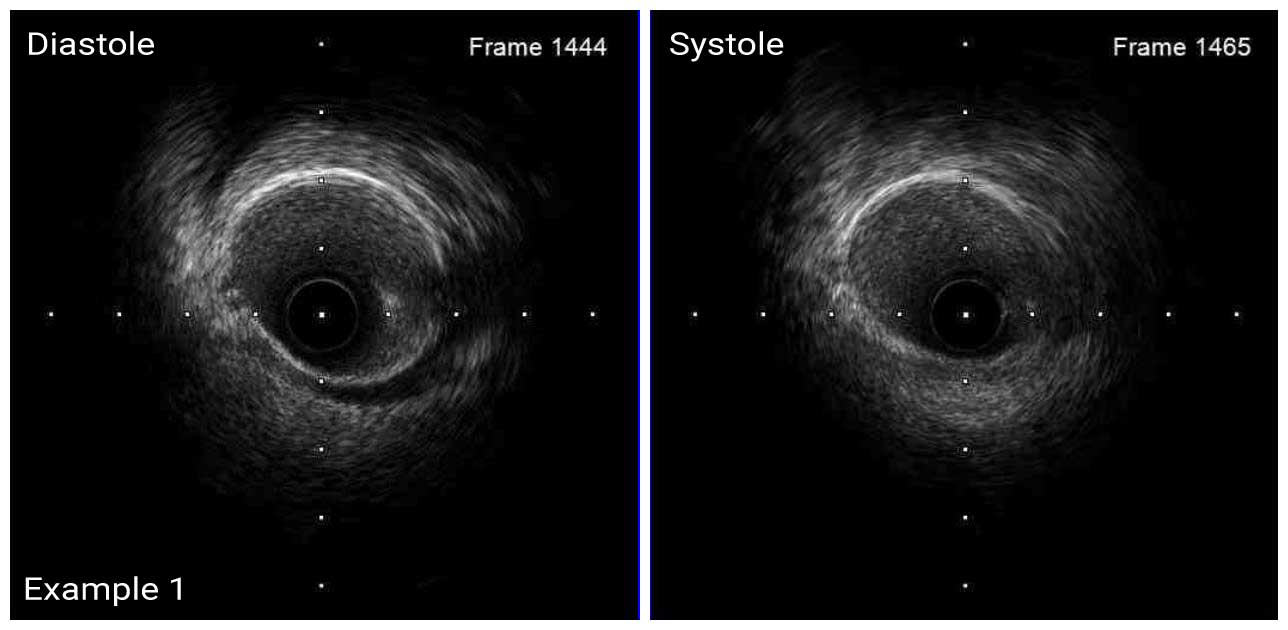
The epicardium appears as a bright layer outside the vessel on IVUS. By identifying the epicardium, the direction of branching of major branching vessels can be inferred. For example, in the LAD, the diagonal branch appears at about 90 degrees counterclockwise of the epicardium. The septal branch appears to be at about 90 degrees clockwise of the epicardium.
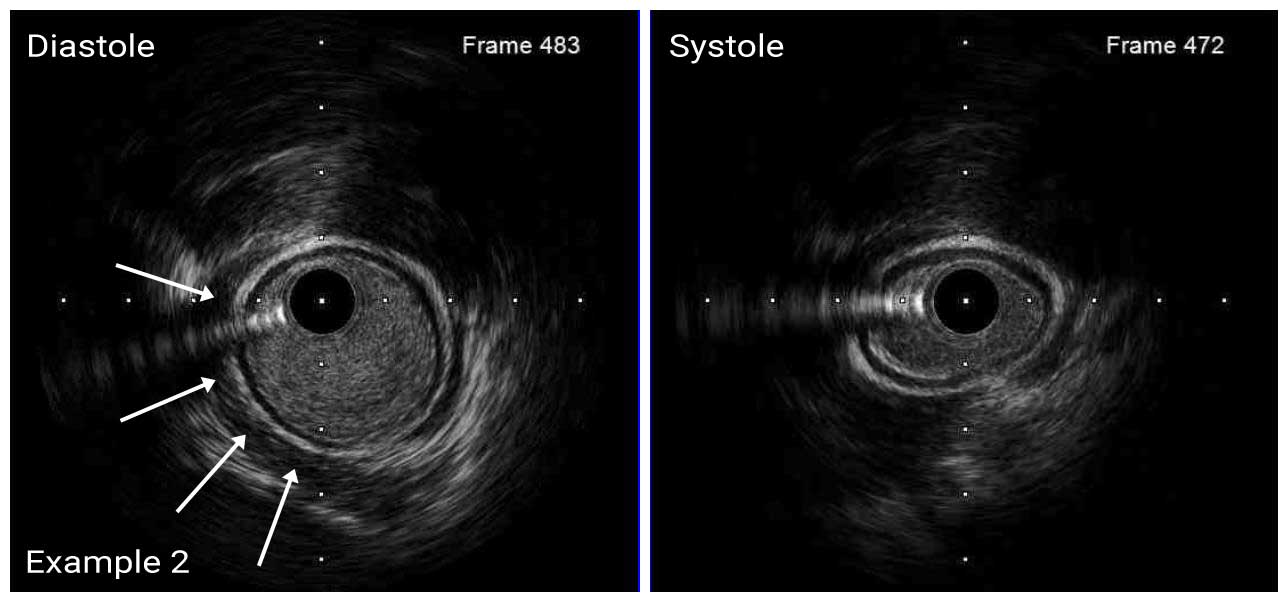
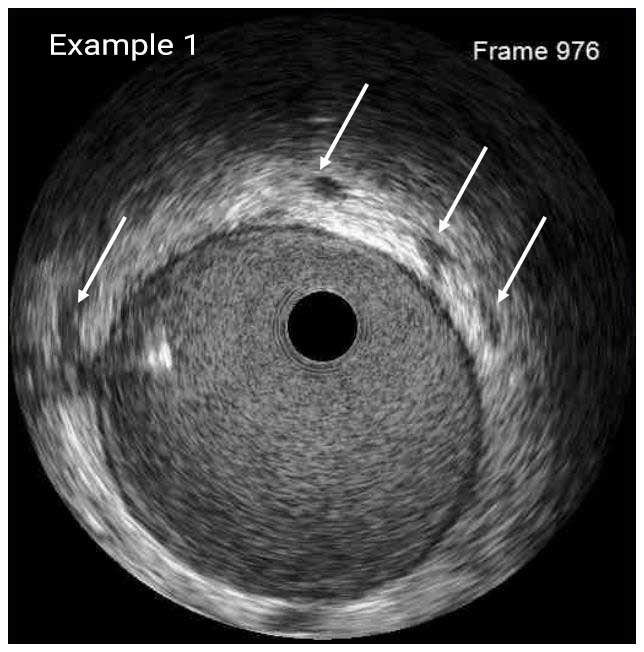
The vasa vasorum of the coronary arteries is a network of small blood vessels that supply the coronary vessel wall.¹ IVUS is able to detect vasa vasorum as a tubular, low-echoic structure at the outer side of a coronary arteries with diameter of several hundred micrometers.²
1. J Am Coll Cardiol 2011; 57: 1961-79
2. JACC Cardiovasc Interv 2013; 6: 985